��Ŀ����
����Ŀ����H2S��CO2�ڸ����·�����Ӧ��H2S(g)+CO2(g)![]() COS(g) +H2O(g)����610 Kʱ����0.10molCO2��0.40molH2S����2.5L�Ŀո�ƿ�У���Ӧƽ���ˮ���������ʵ�������Ϊ0.02����H2S��ƽ��ת����Ϊ____%����Ӧƽ�ⳣ��K=____������С�������λ����
COS(g) +H2O(g)����610 Kʱ����0.10molCO2��0.40molH2S����2.5L�Ŀո�ƿ�У���Ӧƽ���ˮ���������ʵ�������Ϊ0.02����H2S��ƽ��ת����Ϊ____%����Ӧƽ�ⳣ��K=____������С�������λ����
��ijѧ����0.200mol/L�ı�NaOH��Һ�ζ�δ֪Ũ�ȵ����ᣬ������ɷ�Ϊ���¼�����
��������ˮϴ�Ӽ�ʽ�ζ��ܣ���ע��NaOH��Һ����0���̶�������
�ڹ̶��õζ��ܲ�ʹ�ζ��ܼ������Һ��
�۵���Һ������0������0���̶������£������µζ���Һ�����
����ȡ20.00mL����Һע��ྻ����ƿ�У�������3�η�̪��Һ
���ñ�Һ�ζ����յ㣬���µζ���Һ�����
��ش�
��1�����ϲ����д������____�����ţ���
��2���ñ�NaOH��Һ�ζ�ʱ��Ӧ����NaOH��Һע��___��(��ͼ��ѡ����������������)��
��3�����в���������ʵ����ƫ����ǣ�____�����ţ���
A������ƿװҺǰ��������������ˮ
B���ζ�ǰ���ζ��ܼ��������ݣ��ζ���������
C���ζ��յ㸩�Ӷ���
���𰸡�2.5 2.85��10-3 �� �� B
��������
������������ʽ����Ϸ�Ӧƽ���ˮ�����ʵ�������Ϊ0.02����ʽ���㣻
��(1)�ζ��ܵ���0���̶��������̶��ߣ�����ȡ��ʼ������
(2)NaOH��Һ�Ǽ�����Һ���ܹ���������跴Ӧ���ɾ���ճ���ԵĹ����ƣ��ݴ�ѡ��ζ��ܣ�
(3)��c(HCl)=![]() ��֪��V(NaOH)ƫ�ⶨ���ƫ�ݴ˷����жϡ�
��֪��V(NaOH)ƫ�ⶨ���ƫ�ݴ˷����жϡ�
�������ڷ�ӦH2S(g) + CO2(g) COS(g) + H2O(g)
��ʼ(mol) 0.40 0.10 0 0
ת��(mol) x x x x
ƽ��(mol) 0.40-x0.10-x x x
��Ӧƽ���ˮ�����ʵ�������Ϊ0.02����![]() =0.02����ã�x=0.01��H2S��ƽ��ת����a1=
=0.02����ã�x=0.01��H2S��ƽ��ת����a1=![]() ��100%=2.5%��
��100%=2.5%��
���Ϊ2.5L����ƽ��ʱ�����ʵ�Ũ��Ϊc(H2S)=![]() =0.156mol/L��c(CO2)=
=0.156mol/L��c(CO2)=![]() =0.036mol/L��c(COS)=c(H2O)=
=0.036mol/L��c(COS)=c(H2O)=![]() =0.004mol/L����K=
=0.004mol/L����K=![]() =0.00285���ʴ�Ϊ��2.5��0.00285��
=0.00285���ʴ�Ϊ��2.5��0.00285��
��(1)ʵ�鲽���Т�ע��NaOH��Һ����0���̶������ϣ���0���̶��������̶��ߣ�����ȡ�������ʵ����д��ʴ�Ϊ���٣�
(2)�ñ�NaOH��Һ�ζ�ʱ��Ӧ����NaOH��Һע���ʽ�ζ����У������У��ʴ�Ϊ���ң�
(3)A������ƿװҺǰ��������������ˮ����ʵ������Ӱ�죬��A��ѡ��B���ζ�ǰ���ζ��ܼ��������ݣ��ζ��������ݣ��������ĵ�NaOH���ƫ�ⶨ���ƫ��Bѡ��C���ζ����Ӷ�������������NaOH�����ƫС���ⶨ���ƫС����C��ѡ���ʴ�Ϊ��B��

����Ŀ��ijʵ��С����̽��ȩ��������Ӧ��������Һ����ֱ����AgNO3��Һ��ԭ��
��һ����ͬѧ���������¶Ա�ʵ�飺
װ�� | ʵ����� | �Թ��е�ҩƷ | ���� |
| ʵ��� | 2%AgNO3��Һ1mL������2%��ˮ22�Σ��ټ���3���¿�ƿ��40%����ȩ��Һ��ˮԡ����3���� | ���������� |
ʵ��� | 2%AgNO3��Һ1mL������3���¿�ƿ��40%����ȩ��Һ��ˮԡ����3���� | ���������� |
��1����ȩ����������Ӧ�Ļ�ѧ����ʽΪ_____��
��2����ͬѧ��Ϊ��2%AgNO3��ҺӦ�ñ��������Ƶ�������Һ����������ȩ�����ܵ�������_____��д��һ�����ɣ�����ʵ��������������ӻ�ѧ��Ӧԭ���ĽǶȽ��Ϳ��ܵ�ԭ��_____��д��һ�����ɣ���
��������ͬѧΪ���ҵ�������Ӧ��������Һ������ԭ���Ȳ������ϣ�����ȩ������Ӧ�Ļ�����Ϊ����������

��3���ݴˣ�������˸��õ�����ʵ��III��2%AgNO3��Һ1mL������3��1mol/LNaOH��Һ���ټ���2%��ˮ22�Σ��ټ���3���¿�ƿ��40%����ȩ��Һ��ˮԡ���ȣ��ܿ������������
��4���Ҹ���ʵ��III����Ϸ�Ӧ�����г��������ķֲ�������3�����ó��Ľ�����_____��
��������ͬѧΪ��ȷ��AgNO3��Һ������ȩ�������������ϣ��ҵ��˲�ͬŨ�ȵ�AgNO3��Һ������HCHO����ȥ��Ч����ͼ��
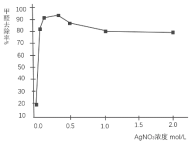
��5����ͼ�пɼ���AgNO3��ҺŨ�ȴﵽһ���̶ȣ�HCHO��ȥ���ʻ������䣬ԭ����AgNO3��ҺŨ��Խ�ߣ�����Խǿ��������______�Ļ�ԭ�ԣ�ͬʱ��˵����AgNO3��Һ��HCHO��Ӧ������������______��
���ģ���ϼ��ұ���ʵ������ϣ���ͬѧ��һ���������Һ��pH�ȶ��������ڿ����γɾ��ȹ�����������
��6�����ϻ�ѧ�������AgNO3��Һ����ȩ��Һ�ķ�ӦpH�仯���ԣ���������Һ�仯������_____��
��7���ۺϸ�ʵ��С����λͬѧ��ʵ�����֤�����ó�������Ӧ��������Һ��������AgNO3��Һ���ŵ���_____��д��2�����ɣ���
����Ŀ����(N2H4)��һ��Ӧ�ù㷺�Ļ���ԭ�ϡ���ҵ���Ⱥϳɰ�����N2(g)+3H2(g)![]() 2NH3(g)��H��0���ٽ�һ���Ʊ��¡����������գ�
2NH3(g)��H��0���ٽ�һ���Ʊ��¡����������գ�

��1���ϳɰ���Ҫѡ����ʵĴ������ֱ�ѡ��A��B��C���ִ����������飬���ý����ͼ��ʾ(����������ͬ)��������������ѡ��Ĵ�����__(����A������B������C��)�������ǣ�__��
��2��һ�������£������ܱ������н��еĺϳɰ���Ӧ��ƽ���������������ʱ����ͬʱѹ������������������¶ȴ���ƽ�����ԭƽ����ȣ��뽫�й��������ı仯����������±���(����������������С��������ȷ����)
��Ӧ���� | ƽ�ⳣ��K | |
�仯��� | ____ | ____ |
��3��ͨ���Ѳ�1molij��ѧ�������յ��������ɸû�ѧ���ļ��ܡ���֪���ֻ�ѧ���ļ������£�
��ѧ�� | N��H | N��N | O==O | N��N | O��H |
����(kJ/mol) | 386 | 167 | 498 | 946 | 460 |
�������۷ɴ��ij������������(N2H4����̬)Ϊȼ�ϣ�����������(��̬)��ȼ�գ�����N2(��̬)��H2O(Һ̬)���÷�Ӧ���Ȼ�ѧ����ʽΪ��____��
��4������Ҫ�����������ʺ����ᡣ��ʮ�������ڼ䣬Ԥ���ҹ��ϳɰ�����������������������ͼ�Dz�ͬ�¶ȺͲ�ͬѹǿ�£���Ӧ�ﵽƽ��������NH3����(���%)�ı仯�������֪��ʼʱn(N2)��n(H2)=1:3���ж�pl��p2ѹǿ�Ĵ�С��ϵ��pl___p2(ѡ��������������������=��)��