��Ŀ����
��������Դ�����ϴ������ǿɳ�����չ����Ҫ���档
��1����̼��������ֱ�Ӻϳ��Ҵ�ȼ���ѽ�����ģ���������ȡ��CO��H2Ϊԭ�Ϻϳ��Ҵ�����ѧ��Ӧ����ʽ��2CO(g)+4H2(g) CH3CH2OH(g)+H2O(g)�����ܱ������г���10 mol CO��20mol H2���ڴ��������·�Ӧ�����Ҵ���CO��ת����(��)���¶ȡ�ѹǿ�Ĺ�ϵ����ͼ��ʾ��
CH3CH2OH(g)+H2O(g)�����ܱ������г���10 mol CO��20mol H2���ڴ��������·�Ӧ�����Ҵ���CO��ת����(��)���¶ȡ�ѹǿ�Ĺ�ϵ����ͼ��ʾ��
����A��B�����ʾ��ijʱ�̴ﵽ��ƽ��״̬����ʱ��A��ʱ���������Ϊ10L������¶��µ�ƽ�ⳣ����K�� ��
����A��C���㶼��ʾ�ﵽ��ƽ��״̬�����Է�Ӧ��ʼ����ƽ��״̬�����ʱ��tA tC������ڡ�����С�ڡ����ڡ�����
�۹�ҵ�ϻ����Բ�ȡ��CO2��H2Ϊԭ�Ϻϳ��Ҵ������Ҹ�����ѧ�������Ƴ磬��������ͬ�����£���CO��ȡCH3CH2OH��ƽ�ⳣ��ԶԶ������CO2��ȡCH3CH2OH��ƽ�ⳣ�������Ʋ⻯ѧ�������Ͽ���CO2��ȡCH3CH2OH���ŵ���Ҫ�ǣ� ��
��2��Ŀǰ��ҵ��Ҳ������CO2�������״���һ�������·�����ӦCO2(g)��3H2(g) CH3OH(g)��H2O(g)������6mol CO2��8 mol H2����2L���ܱ������У����H2�����ʵ�����ʱ��仯����������ͼ��ʾ��ʵ�ߣ���
CH3OH(g)��H2O(g)������6mol CO2��8 mol H2����2L���ܱ������У����H2�����ʵ�����ʱ��仯����������ͼ��ʾ��ʵ�ߣ���
�����ڴ����ͼ�л���״������ʵ�����ʱ��仯���ߡ�
�ڽ��ı�ijһʵ�������ٽ�������ʵ�飬���H2�����ʵ�����ʱ��仯��ͼ��������ʾ������I��Ӧ��ʵ�������ı��� �����ߢ��Ӧ��ʵ�������ı��� ��
��3��Hg��ˮ����Ⱦ���ؽ���Ԫ��֮һ��ˮ��Һ�ж��۹�����Ҫ������̬��Cl����OH����Ũ�ȹ�ϵ����ͼ��ʾ��ͼ�е����ʻ�����ֻ��Hg(OH)2Ϊ�����pCl=��1gc(Cl��)��
������˵������ȷ���� ��
A��Ϊ�˷�ֹHg2��ˮ�⣬����Hg(NO3)2��ҺʱӦ��Hg(NO3)2��������Ũ�������ϡ��
B����c(C1��) ��10��1 mol��L��1ʱ����Ԫ��һ��ȫ����HgCl42����ʽ����
C��HgCl2��һ��������ʣ�����뷽��ʽ�ǣ�HgCl2��HgCl�� + C1��
D������ҺpH������4��pCl��2�ı���6ʱ����ʹHgCl2ת��ΪHg(OH)2
��HgCl2�ֳơ����������۵�549K���������������侧���� ��������ͣ���
��1����K="0.25" ��2�֣� �ڴ��ڣ�2�֣� ��ԭ���á����Լ�������ЧӦ�ȣ�2�֣�
��2���٣�2�֣�
�� ���£� ��ѹ����1�֣�
��3����AD �ڷ��Ӿ���
���������������1���� 2CO(g)+4H2(g) CH3CH2OH(g)+H2O(g)
CH3CH2OH(g)+H2O(g)
��ʼŨ�ȣ�mol/L�� 1 2 0 0
ת��Ũ�ȣ�mol/L��0.5 1.0 0.25 0.25
ƽ��Ũ�ȣ�mol/L��0.5 1.0 0.25 0.25
���ݻ�ѧƽ�ⳣ������һ�������£������淴Ӧ�ﵽƽ��״̬ʱ��������Ũ�ȵ���֮���ͷ�Ӧ��Ũ�ȵ���֮���ı�ֵ��֪�����¶��·�Ӧ��ƽ�ⳣ��K��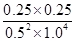 ��0.25��
��0.25��
�ڸ���ͼ���֪��C���¶Ⱥ�ѹǿ������A���¶Ⱥ�ѹǿ������C�㷴Ӧ���ʿ죬�ﵽƽ���ʱ���١�
�����ڵ�����CO2�ĺ������������Ը÷������ŵ���ԭ���á����Լ�������ЧӦ��
��2���ٸ���ͼ���֪ƽ��ʱ���������ʵ�����2mol���������������ʵ�����8mol��2mol��6mol�����Ը��ݷ���ʽ��֪ƽ��ʱ���ɼ״������ʵ�����2mol�����ͼ��ɱ�ʾΪ���𰸡�
������I��ԭ������ȴﵽƽ���ʱ����٣�˵����Ӧ���ʿ졣��ƽ��ʱ���������ʵ������ӣ�˵��ƽ�����淴Ӧ�����ƶ�����������Ӧ�������С�Ŀ��淴Ӧ�����Ըı������ֻ���������¶ȣ�������Ӧ�Ƿ��ȷ�Ӧ�����ߢ���ԭ������ȴﵽƽ���ʱ����٣�˵����Ӧ���ʿ졣��ƽ��ʱ���������ʵ������٣���˵��ƽ��������Ӧ�����ƶ�����������Ӧ�������С�ķ��ȵĿ��淴Ӧ�����Ըı������ֻ��������ѹǿ��
��3����A��Hg2��ˮ����Һ�����ԣ�����Ϊ�˷�ֹHg2��ˮ�⣬����Hg(NO3)2��ҺʱӦ��Hg(NO3)2��������Ũ�������ϡ�ͣ�A��ȷ��B����c(C1��) ��10��1 mol��L��1��pCl��1ʱ����Ԫ����Ҫ����HgCl42����ʽ���ڣ�B����ȷ��C��HgCl2��һ��������ʣ�����뷽��ʽ�ǣ�2HgCl2 HgCl�� + HgCl3����C����ȷ��D������ͼ���֪����ҺpH������4��pCl��2�ı���6ʱ����ʹHgCl2ת��ΪHg(OH)2��D��ȷ����ѡAD��
HgCl�� + HgCl3����C����ȷ��D������ͼ���֪����ҺpH������4��pCl��2�ı���6ʱ����ʹHgCl2ת��ΪHg(OH)2��D��ȷ����ѡAD��
��HgCl2�ֳơ����������۵�549K����������������˵�������Ƿ��Ӿ��塣
���㣺����ƽ�ⳣ���ļ��㡢���������ƽ��״̬��Ӱ���Լ��ܽ�ƽ����й�Ӧ�����ж�

 �¿α�����Ķ�ѵ��ϵ�д�
�¿α�����Ķ�ѵ��ϵ�д� ������������Ӧ����ϵ�д�
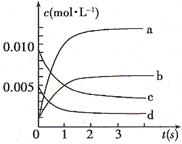
������������Ӧ����ϵ�д���ͼ��ʾij�¶�ʱ����ӦN2O4(g) 2NO2(g)��ǰ110 s�ڵķ�Ӧ��������������ͼ��ش��������⡣
2NO2(g)��ǰ110 s�ڵķ�Ӧ��������������ͼ��ش��������⡣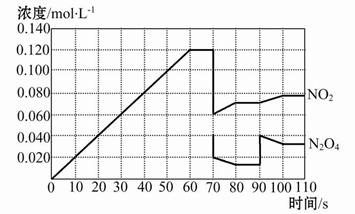
(1)�ÿ��淴Ӧ�ﵽƽ���ʱ���Ϊ ��
(2)��Ӧ���е�70 sʱ���ı������������ (����ĸ����ͬ),��Ӧ���е�90 sʱ���ı������������ ��
| A��������� | B������������� |
| C�������¶� | D������N2O4��Ũ�� |
(4)����ͼ�л�����Ӧ��N2O4��0��70��ʱ��Ũ�ȱ仯���ߡ�
��Դ����������������ٵ��ش���⣬�״���δ����Ҫ����ɫ��Դ֮һ����CH4��H2OΪԭ�ϣ�ͨ�����з�Ӧ���Ʊ��״���
��.CH4��g����H2O��g��=CO��g����3H2��g����H����206.0 kJ/mol
��.CO��g����2H2��g��=CH3OH��g����H����129.0 kJ/mol
��1��CH4��g����H2O��g����Ӧ����CH3OH��g����H2��g�����Ȼ�ѧ����ʽΪ_____��
��2����1.0 mol CH4��2.0 mol H2O��g��ͨ���ݻ�Ϊ100 L�ķ�Ӧ�ң���һ�������·�����Ӧ�����һ����ѹǿ��CH4��ת�������¶ȵĹ�ϵ��ͼ��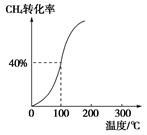
�ټ���100 ��ʱ�ﵽƽ�������ʱ��Ϊ5 min������H2��ʾ�÷�Ӧ��ƽ����Ӧ����Ϊ________��
��100 ��ʱ��Ӧ���ƽ�ⳣ��Ϊ________��
��3����ѹǿΪ0.1 MPa���¶�Ϊ300 �������£���1.0 mol CO��2.0 mol H2�Ļ�������ڴ��������·�����Ӧ�����ɼ״���ƽ����������ݻ�ѹ����ԭ����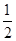 �������������䣬��ƽ����ϵ������Ӱ����________������ĸ��ţ���
�������������䣬��ƽ����ϵ������Ӱ����________������ĸ��ţ���
| A��c��H2����С |
| B������Ӧ���ʼӿ죬�淴Ӧ���ʼ��� |
| C��CH3OH�����ʵ������� |
D������ƽ��ʱ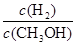 ��С ��С |
��4����ҵ�����ü״��Ʊ������ij��÷����м״��������������÷��е�һ����Ҫ��ӦΪCH3OH��g��
 CO��g����2H2��g�����˷�Ӧ���Է����е�ԭ���ǣ�___��
CO��g����2H2��g�����˷�Ӧ���Է����е�ԭ���ǣ�___����5���״���ˮ�ʻ����һ������Ⱦ����һ�ֵ绯ѧ��������������Ⱦ����ԭ���ǣ�ͨ���Co2��������Co3����Ȼ����Co3������������ˮ�еļ״�������CO2��������ʵ��������ͼװ��ģ���������̣�
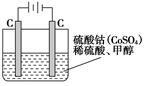
��д�������缫��Ӧʽ_______________��
��д����ȥ�״������ӷ���ʽ___________________��
��2L�ܱ������ڣ�800��ʱ��Ӧ��2NO(g)+O2(g)  2NO2(g)��ϵ�У�n(NO)��ʱ��ı仯�����
2NO2(g)��ϵ�У�n(NO)��ʱ��ı仯�����
| ʱ��(s) | 0 | 1 | 2 | 3 | 4 | 5 |
| n(NO)(mol) | 0.020 | 0.010 | 0.008 | 0.007 | 0.007 | 0.007 |
��1��д���÷�Ӧ��ƽ�ⳣ������ʽ��K= ����֪��
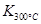 ��
��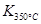 ����÷�Ӧ�� �ȷ�Ӧ��
����÷�Ӧ�� �ȷ�Ӧ����2��ͼ�б�ʾNO2�ı仯�������� ����O2��ʾ��0~2 s�ڸ÷�Ӧ��ƽ������v= ��
��3����˵���÷�Ӧ�Ѵﵽƽ��״̬���� ��
A��v(NO2)=2v(O2)
B��������ѹǿ���ֲ���
C��NO��O2��NO2��Ũ��֮��Ϊ2��1��2
D���������ܶȱ��ֲ���
E���������������ɫ���ٱ仯
��4����С�������ʹѹǿ����ƽ���� ��Ӧ�����ƶ�����������桱����Kֵ �����������С�����䡱��
 2C(g),�����2 s��,����A��ƽ������Ϊ0.06 mol��L-1��s-1,����2 sʱ,�������� mol A,��ʱC�����ʵ���Ũ��Ϊ ��
2C(g),�����2 s��,����A��ƽ������Ϊ0.06 mol��L-1��s-1,����2 sʱ,�������� mol A,��ʱC�����ʵ���Ũ��Ϊ �� 2SO2��g��+ O2��g�� 2SO3��g�������С�H < 0 ������Ӧ�ﵽƽ��ʱ������λ����ͼ2��ʾ��
2SO2��g��+ O2��g�� 2SO3��g�������С�H < 0 ������Ӧ�ﵽƽ��ʱ������λ����ͼ2��ʾ��
 2NH3(g) ��H��0����ʼ �����mol N2����mol H2�� �ﵽƽ���N2��H2��NH3�����ʵ����ֱ�Ϊ1 mol��3 mol��10 mol���ش��������⣺
2NH3(g) ��H��0����ʼ �����mol N2����mol H2�� �ﵽƽ���N2��H2��NH3�����ʵ����ֱ�Ϊ1 mol��3 mol��10 mol���ش��������⣺ 2NH3(g)����H��Q kJ��mol��1�������仯����ͼ���ش��������⣺
2NH3(g)����H��Q kJ��mol��1�������仯����ͼ���ش��������⣺
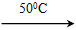 Ni(CO)4(g)��Q
Ni(CO)4(g)��Q Ni(s)��4CO(g)
Ni(s)��4CO(g)