��Ŀ����
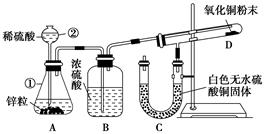
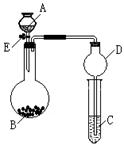
��12�֣�ʵ���ҳ�������װ��������ͭ��Ũ���ᷴӦ��һϵ��ʵ�顣
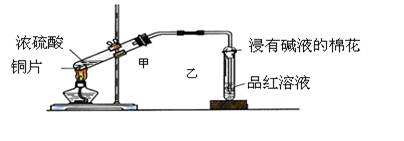
��1������ʲô������ж�ͭ��Ũ���ᷴӦ��SO2����_____________________��
����ʲô������ж�ͭ��Ũ���ᷴӦ������ͭ����______________________��д����װ���з�������Ҫ��Ӧ�Ļ�ѧ����ʽ ��
��2��װ���ҵ��Թܿڲ�����һ�Ž��м�Һ����������ͨ���ǽ��б���̼������Һ���������� _________��
��3��ʵ����Ϻ�ȡ�������Թ�����Һ���Թ��м��ȣ������� ��ԭ���� ��

��1������ʲô������ж�ͭ��Ũ���ᷴӦ��SO2����_____________________��
����ʲô������ж�ͭ��Ũ���ᷴӦ������ͭ����______________________��д����װ���з�������Ҫ��Ӧ�Ļ�ѧ����ʽ ��
��2��װ���ҵ��Թܿڲ�����һ�Ž��м�Һ����������ͨ���ǽ��б���̼������Һ���������� _________��
��3��ʵ����Ϻ�ȡ�������Թ�����Һ���Թ��м��ȣ������� ��ԭ���� ��
(12��)��1������Ʒ���ʳ���ɫ��������Һ����ɫ��
Cu+2H2SO4(Ũ)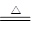 CuSO4+SO2��+2H2O��
CuSO4+SO2��+2H2O��
��2�����ն����SO2��ֹ��Ⱦ���� ��3������ɫ���ɫ��SO2Ư��ʱ���ɲ��ȶ�����ɫ���ʡ�
Cu+2H2SO4(Ũ)
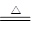 CuSO4+SO2��+2H2O��
CuSO4+SO2��+2H2O����2�����ն����SO2��ֹ��Ⱦ���� ��3������ɫ���ɫ��SO2Ư��ʱ���ɲ��ȶ�����ɫ���ʡ�
��1��SO2����Ư���ԣ���ʹƷ����Һ��ɫ��������Ʒ���ʳ���ɫ��˵����SO2���ɡ�ͭ��������Һ������ɫ���ݴ˿���˵��������ͭ���ɡ�ͭ��Ũ���ᷴӦ�ķ���ʽΪ
Cu+2H2SO4(Ũ)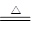 CuSO4+SO2��+2H2O��
CuSO4+SO2��+2H2O��
��2��SO2�ж����Ǵ�����Ⱦ���Ҫβ����������˱���̼���Ƶ����þ������ն����SO2��ֹ��Ⱦ������
��3������SO2��Ư��ԭ���Ǻ���ɫ���ʻ������ɲ��ȶ�����ɫ���ʣ��ڼ��ȵ������£����ָܻ���������ɫ�����Լ���ʱ��Һ�С�����ɫ���ɫ��
Cu+2H2SO4(Ũ)
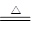 CuSO4+SO2��+2H2O��
CuSO4+SO2��+2H2O����2��SO2�ж����Ǵ�����Ⱦ���Ҫβ����������˱���̼���Ƶ����þ������ն����SO2��ֹ��Ⱦ������
��3������SO2��Ư��ԭ���Ǻ���ɫ���ʻ������ɲ��ȶ�����ɫ���ʣ��ڼ��ȵ������£����ָܻ���������ɫ�����Լ���ʱ��Һ�С�����ɫ���ɫ��

��ϰ��ϵ�д�
 �ܿ�����ĩ��̾�ϵ�д�
�ܿ�����ĩ��̾�ϵ�д�
�����Ŀ