��Ŀ����
����������ĵ���ƽ�ⳣ�����±���
���� | HCOOH | HClO | H2CO3 | H2SO3 |
����ƽ�ⳣ��(25 ��) | Ka��1.77 ��10��4 | Ka��4.0 ��10��8 | Ka1��4.3��10��7 Ka2��4.7��10��11 | Ka1��1.54��10��2 Ka2��1.02��10��7 |
(1)�����¢�0.1 mol��L��1 HCOONa����0.1 mol��L��1 NaClO����0.1 mol��L��1 Na2CO3����0.1 mol��L��1 NaHCO3��Һ��pH�ɴ�С�Ĺ�ϵΪ______________________��
(2)Ũ�Ⱦ�Ϊ0.1 mol��L��1��Na2SO3��Na2CO3�Ļ����Һ�У�SO32-��CO32-��HSO3-��HCO3-Ũ�ȴӴ�С��˳��Ϊ____________________��
(3)����CO2ͨ�뵽������NaClO��Һ�з�����Ӧ�����ӷ���ʽ______________��
(4)�����£�pH��3��HCOOH��Һ��pH��11��NaOH��Һ�������Ϻ���Һ������Ũ���ɴ�С��˳��Ϊ__________________��
����ijС�����ù�ҵ�����ӽ���Ĥ�����ռ��ԭ����������ͼ��ʾװ�õ��K2SO4��Һ��
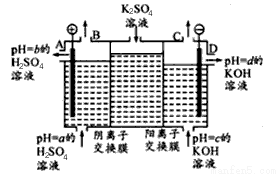
�ٸõ�����ͨ�������ӽ���Ĥ��������_______���>����<����һ����ͨ�������ӽ���Ĥ����������
��ͼ��a��b��c��d�ֱ��ʾ�й���Һ��pH����a��b��c��d��С�����˳��Ϊ _____________��
�۵��һ��ʱ���B������C���ڲ��������������Ϊ______________��



 Ag+(aq)+Cl-(aq)�������м������� NaCl��ĩ��ƽ��������ƶ�����Һ�����ӵ���Ũ�Ȼ��С
Ag+(aq)+Cl-(aq)�������м������� NaCl��ĩ��ƽ��������ƶ�����Һ�����ӵ���Ũ�Ȼ��С