��Ŀ����
6��Ҫ����Ũ��ԼΪ2mol/LNaOH��Һ100mL������IJ�����ȷ���ǣ�������| A�� | ��ȡ8gNaOH���壬����100mL��Ͳ���߽��裬��������������ˮ����������ȫ�ܽ��������ˮϡ����100mL | |
| B�� | ��ȡ8gNaOH���壬����100mL����ƿ�У�������������ˮ��������ƿʹ�����ܽ⣬�ټ���ˮ���̶ȣ��Ǻ�ƿ��������ҡ�� | |
| C�� | ��ȡ8gNaOH�������300mL�ձ��У���100mL��Ͳ��ȡ100mL����ˮ�������ձ��У�ͬʱ���Ͻ����������ܽ� | |
| D�� | ��50mL��Ͳ��ȡ50mL4mol/LNaOH��Һ������100mL�ձ��У�����ͬһ��Ͳȡ50mL����ˮ�����Ͻ����£����������ձ��� |
���� A����Ͳֻ��������ȡҺ�壬���������ܽ���壻
B������ƿֻ������������Һ����������ϡ�ͻ��ܽ�ҩƷ��
C����100mlˮ���ܽ�8gNaOH�������Һ�����ԼΪ100ml�����ǣ�
D������Ͳ��ȡ������������Һ����ϴ��ҺӦ�����Һ�ף�
��� �⣺A����Ͳֻ��������ȡҺ�壬��������ϡ�ͻ��ܽ�ҩƷ����A����
B������ƿֻ������������Һ����������ϡ�ͻ��ܽ�ҩƷ�����������ܽ�ų��������ȣ���������ƿ��ֱ���ܽ����ƣ�Ӱ����Һ�������������ҺŨ�ȱ�����������Σ�գ���B����
C��8g�������Ƶ����ʵ���Ϊ0.2mol������100mlˮ���ܽ�8gNaOH�������Һ�����ԼΪ100ml������Һ��Ũ��ԼΪC=$\frac{0.2mol}{0.1L}$=2mol/L����C��ȷ��
D����50mL��Ͳ��ȡ50mL4mol/LNaOH��Һ������100mL�ձ��У�����ͬһ��Ͳȡ50mLˮ���ᵼ����ȡ���������Ƶ����ʵ���ƫ�࣬��Ũ��ƫ�ߣ���D����
��ѡC��
���� ����������һ�����ʵ���Ũ����ҺΪ���忼����������ʹ�ã��ѶȲ�����ȷ��Ͳֻ��������ȡҺ�壬���������ܽ���壻����ƿֻ������������Һ����������ϡ�ͻ��ܽ�ҩƷ��

��ϰ��ϵ�д�
 �¿α�����Ķ�ѵ��ϵ�д�
�¿α�����Ķ�ѵ��ϵ�д�
�����Ŀ
17���˴Ź���NMR�������ѹ㷺Ӧ���ڸ��ӷ��ӽṹ�IJⶨ��ҽѧ��ϵȸ߿Ƽ�������ֻ֪����������������Ϊ������ԭ�Ӻ���NMR�������ж���������ԭ�Ӿ��ɲ���NMR��������
| A�� | 18O 31P 119Sn | B�� | 27Al 19F 12C | ||
| C�� | 14N 17O 1H | D�� | 13C ${\;}_{20}^{40}Ca$${\;}_{26}^{56}Fe$ |
14������Һ���ܴ��������һ�����ӻ�����ǣ�������
| A�� | NH${\;}_{4}^{+}$��H+��NO${\;}_{3}^{-}$��HCO${\;}_{3}^{-}$ | B�� | K+��Al3+��SO${\;}_{4}^{2-}$��NH3•H2O | ||
| C�� | Na+��K+��SO${\;}_{3}^{2-}$��Cl2 | D�� | Na+��Cl-��CO${\;}_{3}^{2-}$��OH- |
11�������£�������Һ�϶������Ե��ǣ�������
| A�� | ��Һ�к���H+ | B�� | �����̪��Һ����Һ�ʺ�ɫ | ||
| C�� | c��OH-����c��H+������Һ | D�� | ���������Ӧ�ų����� |
15�����л�ѧ������ȷ���ǣ�������
| A�� | �۱�ϩ�Ľṹ��ʽΪ��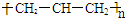 | B�� | ������ӵı���ģ��Ϊ��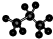 | ||
| C�� | ���Ȼ�̼���ӵĵ���ʽΪ��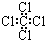 | D�� |  �ķ���ʽ�ǣ�C7H12O �ķ���ʽ�ǣ�C7H12O |
16��X�Ļ�ѧʽΪC8H8����ṹ����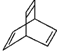 ��ʾ������˵������ȷ���ǣ�������
��ʾ������˵������ȷ���ǣ�������
 ��ʾ������˵������ȷ���ǣ�������
��ʾ������˵������ȷ���ǣ�������| A�� | X������ | |
| B�� | X��ʹ���Ը��������Һ��ɫ | |
| C�� | X��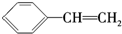 ��Ϊͬ���칹�� ��Ϊͬ���칹�� | |
| D�� | X��������H2��һ�������·�Ӧ�����ɻ�״�ı�����Z��Z��һ�ȴ�����4�� |
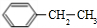 ��
�� ��
�� ��
�� ��
��