��Ŀ����
 ij���������Ҫ��Ϊ�������ж������������ó���0.02mg?L-1��ijͬѧ������ͼ��ʾ����װ�òⶨ�����е�SO2������ȷ��ȡ10mL 5��10-4mol?L-1�ı���ˮ��Һ��ע���Թ��У���2��3�ε���ָʾ������ʱ��Һ����ɫ����ָ���IJⶨ�ص������ÿ�γ���100mL��ֱ����Һ����ɫȫ���ʾ�Ϊֹ�������ͬѧ�IJ�����ȷ�ģ����������Ĵ�������Ϊ
ij���������Ҫ��Ϊ�������ж������������ó���0.02mg?L-1��ijͬѧ������ͼ��ʾ����װ�òⶨ�����е�SO2������ȷ��ȡ10mL 5��10-4mol?L-1�ı���ˮ��Һ��ע���Թ��У���2��3�ε���ָʾ������ʱ��Һ����ɫ����ָ���IJⶨ�ص������ÿ�γ���100mL��ֱ����Һ����ɫȫ���ʾ�Ϊֹ�������ͬѧ�IJ�����ȷ�ģ����������Ĵ�������Ϊ���㣺̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ����,Ԫ�ؼ��仯����
���������������õ���Һ�����գ�����ʽΪ��SO2+I2+2H2O�T2HI+H2SO4����Ϸ�Ӧ�ķ���ʽ���㣮
���
�⣺���������õ���Һ�����գ�����ʽΪ��SO2+I2+2H2O�T2HI+H2SO4��Ϊ˵���õؿ����е�SO2���������ŷű���
�����ٳ���x�Σ������x��SO2������Ϊ100x��10-3L��2��10-5 g?L-1=��2x��10-6��g������ݻ�ѧ����ʽ�ɵó����ۣ�
SO2����������������������I2
64 g��������������������1 mol
2x��10-6 g����10��10-3 L��5��10-4 mol?L-1
x=160��
�ʴ�Ϊ��160��
�����ٳ���x�Σ������x��SO2������Ϊ100x��10-3L��2��10-5 g?L-1=��2x��10-6��g������ݻ�ѧ����ʽ�ɵó����ۣ�
SO2����������������������I2
64 g��������������������1 mol
2x��10-6 g����10��10-3 L��5��10-4 mol?L-1
x=160��
�ʴ�Ϊ��160��
���������⿼�����ʺ����IJⶨ��Ϊ��Ƶ���㣬���շ����Ļ�ѧ��ӦΪ���Ĺؼ���ע�����ù�ϵʽ������㣬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ѧϰʵ����ϵ�д�
ѧϰʵ����ϵ�д�
�����Ŀ
 �ݱ����������⻯����NaBH4��BԪ�صĻ��ϼ�Ϊ+3�ۣ���H2O2��ԭ�ϵ�ȼ�ϵ�أ��������ϲ���Pt/C���������ϲ���MnO2���������վ�ͨ�����ǵ�Դ���乤��ԭ����ͼ��ʾ������˵����ȷ���ǣ�������
�ݱ����������⻯����NaBH4��BԪ�صĻ��ϼ�Ϊ+3�ۣ���H2O2��ԭ�ϵ�ȼ�ϵ�أ��������ϲ���Pt/C���������ϲ���MnO2���������վ�ͨ�����ǵ�Դ���乤��ԭ����ͼ��ʾ������˵����ȷ���ǣ�������| A����طŵ�ʱNa+��b��������a���� |
| B���缫b����MnO2��MnO2�����缫�������д����� |
| C���õ�صĸ�����ӦΪ��BH4-+8OH-+8e-��BO2-+6H2O |
| D��ÿ����3mol H2O2��ת�Ƶĵ���Ϊ3mol |
�й�������;������˵������ȷ�ǣ�������
| A��������Ũ������ʹ�������ۻ������ڳ�����������������������Ũ���� |
| B���γ����Ͻ��˷��˽�������ǿ�Ƚϵͺ���ĥ�Խϲ�IJ��� |
| C������������������ʢװ������Һ��������ʢװǿ������Һ |
| D�����ڸ������ܻ�ԭ�̡��������Ƚ���������õ����� |
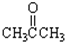 ����CH3CH2Br����
����CH3CH2Br����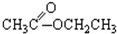 ����
����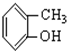 ����
����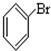 ����
����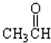 ����
����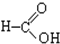 ����
����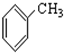 ����
����