��Ŀ����
��14�֣���֪��AΪ���������ӵĵ���ɫ���廯̨�E��XΪ�����г������壬A��B��C��D������ͬ�Ľ������ӣ���ת����ϵ����ͼ�����ֲ�������ȥ����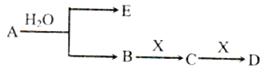
��ش���������
��1�����ֽ������ӵ����ӽṹʾ��ͼΪ_____________;
��2��X�ĵ���ʽ_______________;
��3��B��������ѧ����������_____________;
���³�ѹ�£�7.8gA��������ˮ��ַ�Ӧ�ų�����a kJ��д���÷�Ӧ���Ȼ�ѧ����ʽ
_________________________________________________________________.
��4����CҲ��ת��ΪB��д����ת���Ļ�ѧ����ʽ_______________________________;
��B��D��Ӧ�����ӷ���ʽΪ_______________________________________.
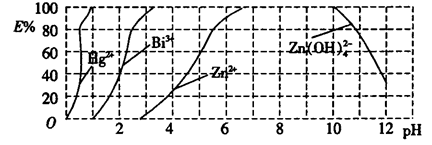
��5����һ����������Xͨ��2LB����Һ�У���������Һ�б���μ���ϡ��������������� �������������������ʵ����Ĺ�ϵ��ͼ������������ܽ��HCl�Ļӷ�����
��ش�a����Һ���������ʵĻ�ѧʽΪ__________ ��b����Һ�и�����Ũ���ɴ�С�Ĺ�ϵ��_________________________________��
��14�֣�
��1��Na+ (1��)
(1��)
��2�� (2��)
(2��)
��3�������ԣ����ۼ������Ӽ���2�֣�2Na2O2(s)+2H2O(l)=4NaOH(aq)+ O2(g)����H=-20akJ/mol(2��)
��4����Ca(OH)2+Na2CO3=CaCO3��+2NaOH�� Ba(OH)2+Na2CO3=BaCO3��+2NaOH
(2��)
��OH-+HCO3-=CO32-+ H2O(2��)
��5��Na2CO3��NaCl (2��)
c(Na+)>c(Cl-)>c(HCO3-)> c(OH-)> c(H+)> c(OH-)> c(CO32-)( c(CO32-)��д�ɲ�д��������������)��2�֣�
���������������1��AΪ���������ӵĵ���ɫ���廯�����AΪNa2O2������������Na+�����ӽṹʾ��ͼΪ ��
��
��2��A��B��C��D������ͬ�Ľ������ӣ���BΪNaOH��CΪNa2CO3��DΪNaHCO3��XΪCO2�������ʽΪ ��
��
��3��NaOH�к������Ӽ����ۼ���
��4����Na2CO3ת��ΪNaOH���������ֽⷴӦ����Ҫ�Լ��������ƻ�������������ѧ����ʽΪCa(OH)2+Na2CO3=CaCO3��+2NaOH��Ba(OH)2+Na2CO3=BaCO3��+2NaOH��
��NaOH��NaHCO3��Ӧ����̼���ƺ�ˮ�����ӷ���ʽΪOH-+HCO3-=CO32-+ H2O��
��5����ͼ��֪����������̼������2������ᣬ����������̼֮ǰ��3������ᣬ��Na2CO3��NaHCO3��Ҫ��������NaHCO3��CO2��Ҫ�����������ͬ��˵���μ�����ǰ����Һ�е�����ΪNa2CO3��NaOH�������ʵ���֮��Ϊ2:1������a��֮ǰ�����ķ�Ӧ���������������Ƶ��кͷ�Ӧ��a�������ΪNa2CO3��NaCl��b��ʱNa2CO3ȫ��ת��ΪNaHCO3����ʱ����ΪNaHCO3��NaCl�������ʵ���֮��Ϊ2:3��������Һ�е�����Ũ�ȴ�С��ϵΪc(Na+)>c(Cl-)>c(HCO3-)> c(OH-)> c(H+)> c(OH-)> c(CO32-)��
���㣺�������ӽṹʾ��ͼ������ʽ����ѧ����ʽ�����ӷ���ʽ���ж�����д������Ũ�ȴ�С�ıȽ�

��M��N������Һ�����ⶨ��������Һ�к�������12�����ӣ�Al3����Cl����Na����K����NO3-��OH����Fe2����AlO2-��CO32-��NH4+��SO42-��H����
(1)������б�����ʵ��ٵĽ��ۺ�ʵ��ڵ�ʵ�������Լ�����
| ʵ�������Լ����� | ���� |
| ��ȡ����N��Һ�μ����������ᱵ��Һ���������� | N�в��� ���� |
| �� | ȷ��M��Һ�к���Na��������K�� |
 ����pH��ֽ���M��Һ��pH��ֽ����ɫ ����pH��ֽ���M��Һ��pH��ֽ����ɫ | |
NO3-������________��Һ�У�������____________________________________��
Cl��������________��Һ�У��Ǹ���_________________ ȷ���ġ�
(3)����(1)�е�ʵ��ȷ����M��Һ�к��е�����Ϊ________________________��
�������ȣ�ClO2��Ϊһ�ֻ���ɫ���壬�ǹ����Ϲ��ϵĸ�Ч�����ס����١���ȫ��ɱ����������
��1����ҵ���Ʊ�ClO2�ķ�Ӧԭ�������ã�2NaClO3��4HCl=2ClO2����Cl2����2H2O��2NaCl��
�� Ũ�����ڷ�Ӧ����ʾ������������_______������ţ���
| A��ֻ�л�ԭ�� | B����ԭ�Ժ����� | C��ֻ�������� | D�������Ժ����� |
��2��Ŀǰ�ѿ������õ�ⷨ��ȡClO2���¹��ա�
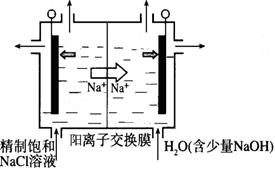
����ͼʾ����ʯī���缫����һ�������µ�ⱥ��ʳ��ˮ��ȡClO2��д����������ClO2�ĵ缫��Ӧʽ��__________��
�ڵ��һ��ʱ�䣬�������������������Ϊ112 mL����״����ʱ��ֹͣ��⡣ͨ�������ӽ���Ĥ�������ӵ����ʵ���Ϊ_________mol����ƽ���ƶ�ԭ������������pH�����ԭ��______________��
��3��ClO2����ˮ��Fe2+��Mn2+��S2����CN-�������Ե�ȥ��Ч����ij������ˮ�к�CN- a mg/L������ClO2��CN-������ֻ�����������壬�����ӷ�Ӧ����ʽΪ_______������100 m3������ˮ��������ҪClO2 _______ mol ��
ʳ���к���һ������þ���������ʣ��ӵ����е����ʧ��Ҫ���������ʡ�ˮ�֡������е������Լ����ա����ȶ�����ġ���֪��
�����ԣ�IO3-��Fe3����I2����ԭ�ԣ�S2O32-��I��
3I2��6OH��=IO3-��5I����3H2O��KI��I2 KI3
KI3
(1)ijѧϰС��Լӵ��ν�������ʵ�飺ȡһ����ij�ӵ���(���ܺ���KIO3��KI��Mg2����Fe3��)������������ˮ�ܽ⣬����ϡ�����ữ����������Һ��Ϊ3�ݡ���һ����Һ�еμ�KSCN��Һ���Ժ�ɫ���ڶ�����Һ�м�����KI���壬��Һ�Ե���ɫ����CCl4��ȡ���²���Һ���Ϻ�ɫ����������Һ�м�������KIO3����μӵ����Լ�����Һ����ɫ��
�ټ�KSCN��Һ���Ժ�ɫ���ú�ɫ������_____________(�û�ѧʽ��ʾ)��CCl4�����Ϻ�ɫ��������___________(�õ���ʽ��ʾ)��
�ڵڶ�����Һ�м�������KI�����Ӧ�����ӷ���ʽΪ_______________________________________��__________________________________________________________________��
(2)KI��Ϊ�ӵ����ʳ���ڱ�������У����ڿ��������������ã�������������ʧ��
д����ʪ������KI��������Ӧ�Ļ�ѧ����ʽ��_____________________��
��I2����KI��Һ���ڵ��������£����Ƶ�KI3��H2O����������Ϊʳ�μӵ���Ƿ���ʣ�___________ (��ǡ���)����˵������_________________________________________________��
(3)Ϊ����ӵ���(����KI)���ȶ��ԣ��ɼ��ȶ������ٵ����ʧ�������������п�����Ϊ�ȶ�������_______��
| A��Na2S2O3 | B��AlCl3 | C��Na2CO3 | D��NaNO2 |
ijԪ���ڻ�ѧ��Ӧ���ɻ���̬��Ϊ����̬�����Ԫ��
| A��һ���������ˡ������� | B��һ������ԭ�� |
| C���ȿ��ܱ���ԭҲ���ܱ����� | D���Ȳ����ܱ������ֲ����ܱ���ԭ |
����ɫ��ѧ���������Ի�������Ⱦ�����������п��Գ�Ϊ����ɫ������������
| A��Ũ���� | B������ | C��˫��ˮ | D��Ư�� |
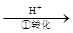 Cr2O72��
Cr2O72��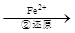 Cr3+
Cr3+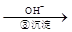 Cr(OH)3��
Cr(OH)3�� Cr2O72��(��ɫ)+H2O
Cr2O72��(��ɫ)+H2O Cu (HDZ)2+2H+���ټ���CCl4��Cu (HDZ)2�ͺ����ױ���ȡ��CCl4�С�
Cu (HDZ)2+2H+���ټ���CCl4��Cu (HDZ)2�ͺ����ױ���ȡ��CCl4�С�