��Ŀ����
��14�֣�ijѧϰС����ͨ����ӦNa2S2O3+H2SO4=Na2SO4+S��+SO2��+H2O�о���Ӧ���ʵ�Ӱ�����غ�Na2S2O3��������Ȥ��������֪Na2S2O3����Ϊ��������ƣ��׳ƺ��������Կ�������һ��Sԭ��ȡ����Na2SO4�е�һ��Oԭ�Ӷ��γɡ���ʵ��С���������ѧϰ��˼��Ԥ����Na2S2O3��ijЩ���ʣ���ͨ��ʵ��̽����֤�Լ���Ԥ�⡣
[�������]
��1������ѧ����ΪNa2S2O3��Na2SO4�ṹ���ƣ���ѧ����Ҳ���ƣ��������ʱNa2S2O3��Һ��pH 7���>������=����<����
��2������ѧ����SԪ�ػ��ϼ��Ʋ�Na2S2O3��SO2�������ƣ������н�ǿ�� ��
[ʵ��̽��]
ȡ����Na2S2O3���壬����ˮ���Ƴ�Na2S2O3��Һ����������̽������д�±��пո�
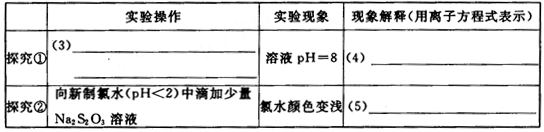
[ʵ�����]
��6��̽���٣� ��
��7��̽���ڣ� ��
[��������]
��8����ͬѧ��̽���ڡ���Ӧ�����Һ�еμ���������Һ���۲쵽�а�ɫ�������������ݴ���Ϊ��ˮ�ɽ�Na2S2O3����������Ϊ�÷����Ƿ���ȷ��˵������
��
��9�������������һ��ʵ�鷽����֤��Na2S2O3����ˮ��������ķ�����
��
[�������]
��1������ѧ����ΪNa2S2O3��Na2SO4�ṹ���ƣ���ѧ����Ҳ���ƣ��������ʱNa2S2O3��Һ��pH 7���>������=����<����
��2������ѧ����SԪ�ػ��ϼ��Ʋ�Na2S2O3��SO2�������ƣ������н�ǿ�� ��
[ʵ��̽��]
ȡ����Na2S2O3���壬����ˮ���Ƴ�Na2S2O3��Һ����������̽������д�±��пո�

[ʵ�����]
��6��̽���٣� ��
��7��̽���ڣ� ��
[��������]
��8����ͬѧ��̽���ڡ���Ӧ�����Һ�еμ���������Һ���۲쵽�а�ɫ�������������ݴ���Ϊ��ˮ�ɽ�Na2S2O3����������Ϊ�÷����Ƿ���ȷ��˵������
��
��9�������������һ��ʵ�鷽����֤��Na2S2O3����ˮ��������ķ�����
��

(1)=
(2)��ԭ��
(3)�ò�����պȡ����ҺNa2S2O3����pH��ֽ�ϣ�Ȼ�������ɫ���Ƚϣ�ȷ����pHֵ
(4)S2O32�D��H2O
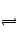 HS2O3�D��OH�D,�������������һ�����൱���������е��������ˮ�������������ˮ��Һ�����ԡ�
HS2O3�D��OH�D,�������������һ�����൱���������е��������ˮ�������������ˮ��Һ�����ԡ�(5)��������ư�Cl2��ԭ:S2O32�D��4Cl2��5H2O=2SO42�D��8Cl�D��10H��
(6)���������ˮ��Һ�ʼ��ԡ�
(7)��������ƾ��л�ԭ��
(8)�÷���������������ˮ������Ag����Cl�D������AgCl������
(9)ȡ��Ӧ�����Һ�����������Ȼ�����Һ���ټ������ᣬ�������ij������ܽ⣬����������Ʊ������ˣ�����û��������

��ϰ��ϵ�д�
�����Ŀ
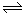 2C��g������2�����C��Ũ��Ϊ0.6 mol?L-1��������
2C��g������2�����C��Ũ��Ϊ0.6 mol?L-1��������
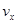 ����x����ij��Ӧ��������֮��Ĺ�ϵ����ȷ���ǣ� ��
����x����ij��Ӧ��������֮��Ĺ�ϵ����ȷ���ǣ� ��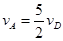 ��
��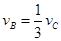 ��
��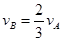 ��
��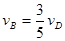
 4NO��6H2O��5 L���ܱ������н��У�����Ӻ�NO�����ʵ���������0.3 mol����˷�Ӧ��ƽ������
4NO��6H2O��5 L���ܱ������н��У�����Ӻ�NO�����ʵ���������0.3 mol����˷�Ӧ��ƽ������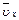 (��ʾ��Ӧ����������ʻ����������������)Ϊ�� ��
(��ʾ��Ӧ����������ʻ����������������)Ϊ�� �� 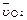 �� 0.01 mol / (L��s)
�� 0.01 mol / (L��s) �� 0.008 mol / (L��s)
�� 0.008 mol / (L��s)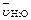 �� 0.003 mol / (L��s)
�� 0.003 mol / (L��s) �� 0.004 mol / (L��s)
�� 0.004 mol / (L��s)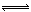 4NO��g����6H2O��g������������ȷ����
4NO��g����6H2O��g������������ȷ���� ��
��