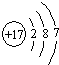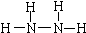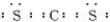��Ŀ����
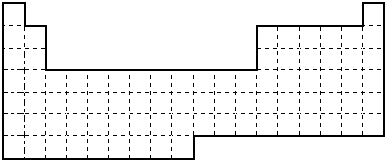
X��Y��V��W�dz����Ķ�����Ԫ�أ����������ڱ����λ������ͼ��ʾ��V��W�������̬�⻯��M��N���ʱ�а������ɣ�M�Ǽ������壮��ش��������⣺
��1��W��ԭ�ӽṹʾ��ͼΪ______��
��2�����³�ѹ�£���a L M�����b L N����ͨ��ˮ�У���������Һ��pH=7����a______b�������=��������
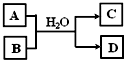
��3���������ѣ�TiO2����X�ĵ��ʡ�Y�ĵ����ڸ����·�Ӧ������������Ԫ����ɵ����ֻ�����÷�Ӧ�Ļ�ѧ����ʽΪ______��
��4��������N2H4��Ҳ���£��ǻ����ȼ��д���µĽṹʽ______���ô���������Һ����M�ܵõ��µ�ϡ��Һ��д����Ӧ�����ӷ���ʽ______��
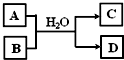
��5����XԪ�ص�һ�ֻ�����A��Y�����������B֮������ͼ��ת����ϵ����DΪ��ɫ����������ͼת�������ӷ���ʽΪ______��
| Y | V | |||
| X | W |
��2�����³�ѹ�£���a L M�����b L N����ͨ��ˮ�У���������Һ��pH=7����a______b�������=��������
��3���������ѣ�TiO2����X�ĵ��ʡ�Y�ĵ����ڸ����·�Ӧ������������Ԫ����ɵ����ֻ�����÷�Ӧ�Ļ�ѧ����ʽΪ______��
��4��������N2H4��Ҳ���£��ǻ����ȼ��д���µĽṹʽ______���ô���������Һ����M�ܵõ��µ�ϡ��Һ��д����Ӧ�����ӷ���ʽ______��
��5����XԪ�ص�һ�ֻ�����A��Y�����������B֮������ͼ��ת����ϵ����DΪ��ɫ����������ͼת�������ӷ���ʽΪ______��

V��W�������̬�⻯��M��N���ʱ�а������ɣ�M�Ǽ������壬��MΪNH3��NΪHCl������VΪNԪ�أ�WΪClԪ�أ���������Ԫ�������ڱ��е�λ�ÿ�֪��YΪCԪ�أ�XΪAlԪ�أ���
��1��WΪClԪ�أ�ԭ������Ϊ17��ԭ�Ӻ�����3�����Ӳ㣬����������Ϊ7����ԭ�ӽṹʾ��ͼΪ
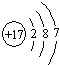
��
�ʴ�Ϊ��
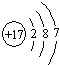
��
��2��MΪNH3��NΪHCl������ߵ����ʵ���������ǡ�÷�Ӧ����NH4Cl��Ϊǿ�������Σ���Һ�����ԣ���������Һ��pH=7������Ӧ�������ʴ�Ϊ������
��3��YΪCԪ�أ�XΪAlԪ�أ���������ѣ�TiO2���ڸ����·�Ӧ���ɵ����ֻ�����ΪAl2O3��TiC����Ӧ�Ļ�ѧ����ʽΪ3TiO2+4Al+3C
2Al2O3+3TiC��
�ʴ�Ϊ��3TiO2+4Al+3C
2Al2O3+3TiC��
��4��������N2H4����Nԭ������������ԭ�ӳɼ����ﵽ8�����ȶ��ṹ���ṹʽΪ
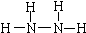
������������Һ����NH3�ܵõ��µ����ӷ���ʽΪClO-+2NH3=N2H4+Cl-+H2O��
�ʴ�Ϊ��
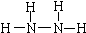
��ClO-+2NH3=N2H4+Cl-+H2O��
��5����XԪ�ص�һ�ֻ�����A��Y�����������B��CO2��֮�䷢����Ӧ�����ɰ�ɫ����DΪAl��OH��3��ΪAlO2-��Һ��CO2�ķ�Ӧ����Ӧ�����ӷ���ʽΪAlO2-+CO2+2H2O=Al��OH��3��+HCO3-��2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��
�ʴ�Ϊ��AlO2-+CO2+2H2O=Al��OH��3��+HCO3-��2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��
��1��WΪClԪ�أ�ԭ������Ϊ17��ԭ�Ӻ�����3�����Ӳ㣬����������Ϊ7����ԭ�ӽṹʾ��ͼΪ
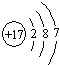
��
�ʴ�Ϊ��
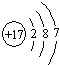
��
��2��MΪNH3��NΪHCl������ߵ����ʵ���������ǡ�÷�Ӧ����NH4Cl��Ϊǿ�������Σ���Һ�����ԣ���������Һ��pH=7������Ӧ�������ʴ�Ϊ������
��3��YΪCԪ�أ�XΪAlԪ�أ���������ѣ�TiO2���ڸ����·�Ӧ���ɵ����ֻ�����ΪAl2O3��TiC����Ӧ�Ļ�ѧ����ʽΪ3TiO2+4Al+3C
| ||
�ʴ�Ϊ��3TiO2+4Al+3C
| ||
��4��������N2H4����Nԭ������������ԭ�ӳɼ����ﵽ8�����ȶ��ṹ���ṹʽΪ
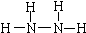
������������Һ����NH3�ܵõ��µ����ӷ���ʽΪClO-+2NH3=N2H4+Cl-+H2O��
�ʴ�Ϊ��
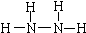
��ClO-+2NH3=N2H4+Cl-+H2O��
��5����XԪ�ص�һ�ֻ�����A��Y�����������B��CO2��֮�䷢����Ӧ�����ɰ�ɫ����DΪAl��OH��3��ΪAlO2-��Һ��CO2�ķ�Ӧ����Ӧ�����ӷ���ʽΪAlO2-+CO2+2H2O=Al��OH��3��+HCO3-��2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��
�ʴ�Ϊ��AlO2-+CO2+2H2O=Al��OH��3��+HCO3-��2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��

��ϰ��ϵ�д�
 53���ò�ϵ�д�
53���ò�ϵ�д�
�����Ŀ