��Ŀ����
����˵������֤�����淴ӦN2��3H2 2NH3�Ѵﵽƽ��״̬����
2NH3�Ѵﵽƽ��״̬����
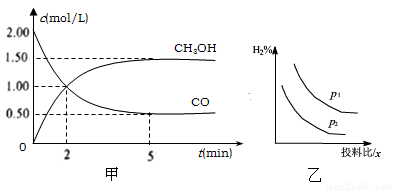
��һ��N��N�����ѵ� ͬʱ����6��N��H������
ͬʱ����6��N��H������
��v(H2)��0.6 mol��L��1��min��1��v(NH3)��0.4 mol��L��1��min��1
�۱���������������ʱ����ϵѹǿ���ٸı�
��NH3��N2��H2��������������ٸı�
�ݺ��º���ʱ����������������ֲ���
A. �ڢۢ� B. �٢ۢ� C. �٢ڢ� D. �ۢܢ�
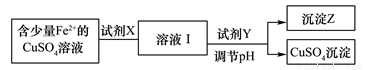
�����ڱ��������ڵڢ���Ԫ�أ��仯������;�㷺���磺LiCoO2��﮵�ص��������ϣ������ܿ�����ָʾ���ʹ����Ʊ���
��.��1��LiCoO2����Ԫ�صĻ��ϼ�Ϊ_______
��2����ҵ�Ͻ���﮵�ص��������������������Һ��ϡ�����ϼ��ȣ��ɵõ�CoSO4���գ���Ӧ�Ļ�ѧ����ʽΪ��______________�������������H2SO4��H2O2�Ļ��Һ����ȱ����____________
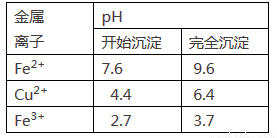
��.����һ�ֺ��ܿ�ʯ[��Ҫ�ɷ�ΪCo2O3��������Fe2O3��Al2O3��MnO��MgO��CaO��]��ȡCoC2O4��2H2O�����������£�
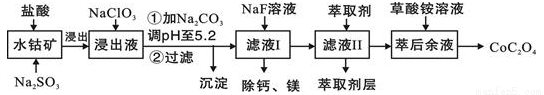
��֪���ٽ���Һ���е���������Ҫ��H+��Co2+��Fe2+��Mn2+��Ca2+��Mg2+��Al3+�ȣ�
�ڲ���������������������ʽ����ʱ��Һ��pH���±���
������ | Fe(OH)3 | Fe(OH��2 | Co(OH)2 | Al(OH)3 | Mn(OH)2 |
��ȫ���� | 3.7 | 9.6 | 9.2 | 5.2 | 9.8 |
��3������Һ�м�NaClO3��Ŀ���ǣ�______________��
��4������ƽ���ƶ�ԭ��˵����Na2CO3��pH��5.2���ó�����ԭ��____________
��5����Һ���м�����ȡ����������___________
��6�������ơ�þ���ǽ���Һ��Ca2+��Mg2+ת��ΪMgF2��CaF2��������֪Ksp(MgF2)��7.35��10-11��Ksp(CaF2)��1.05��10-10�����������NaF��������Һc(Mg2+)/c(Ca2+)��___________��


 2CO2(g)��S(s) ��H����a kJ/mol(a��0)������������ȷ����
2CO2(g)��S(s) ��H����a kJ/mol(a��0)������������ȷ����
 2NH3(g) ��H���÷�Ӧ��һ�����������Է��������й��ڸ÷�Ӧ�Ħ�H����S���жϣ���ȷ����
2NH3(g) ��H���÷�Ӧ��һ�����������Է��������й��ڸ÷�Ӧ�Ħ�H����S���жϣ���ȷ���� CH3OH(g) ��H��
CH3OH(g) ��H��