��Ŀ����
��14�֣�
����һ�������µ����п��淴Ӧ�ﵽƽ��ʱ���������xA��y Bz C
��1����A��B��C�������壬�ڼ�ѹ��ƽ�����淴Ӧ�����ƶ�����x��y��z��ϵ��________��
��2����C�����壬����x+y=z���ڼ�ѹʱ��ѧƽ��ɷ����ƶ�����ƽ��ض�����_____�����ƶ���
��3����֪B��C�����壬������A���ʵ�����ƽ�ⲻ�ƶ���˵��A��______����״̬����
��4������Ⱥ�C�İٷֺ�����С��������Ӧ��________�ȷ�Ӧ��
����ͼ��ʾ��A�г���1 mol X ��1 mol Y����B�г���2 mol X��2 mol Y����ʼʱ��V��A��=V��B��= a L������ͬ�¶Ⱥ��д��������£��������и��Է���������Ӧ��X(g)+Y(g)2Z(g)+W(g)����H��0���ﵽƽ��ʱ��V��A��= 1.2a L��
�Իش�
��1��A��X��ת���ʦ�(A)___________________________��
��2��A��B��X��ת���ʵĹ�ϵ����(A) ___________��(B)��(�������������=��)
��3����K��һ��ʱ���ִﵽƽ�⣬A�����Ϊ___________L������ͨ��������������ƣ�
��1��x+y��z��2���淴Ӧ����(�������ƶ�)(3���̻�Һ����4������
��(1)40% (2)�� (3)2.6a
����:

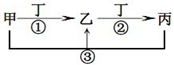 �ס��ҡ�����������ѧ���������ʣ����мס��ҡ���������ͬһ��Ԫ�أ���һ�������µ�ת����ϵ��ͼ������˵����ȷ���ǣ�������
�ס��ҡ�����������ѧ���������ʣ����мס��ҡ���������ͬһ��Ԫ�أ���һ�������µ�ת����ϵ��ͼ������˵����ȷ���ǣ�������| A������Ϊ���������;���Ľ������ʣ��ҵ���Һһ��ΪFeCl3 | ||||
B����ͨ������¼ס��ҡ��������������壬���ҺͶ�Ϊ��������Ҫ�ɷ֣���Ӧ�ٵĻ�ѧ����ʽ4NH3+5O2
| ||||
| C�����ס��ҡ�������Һ�Լ��ԣ�������Ϊҽ��������θ�����֢��ҩ�����������ʵ������Һͱ�����ˮ�γɻ����Һ����Һ�и�����Ũ���ɴ�С��˳��Ϊc��Na+����c��HCO3-����c��CO32-����c��OH-����c��H+�� | ||||
| D������Ϊ�������Ϊ�ȼҵ����Ҫ��Ʒ�����һ��Ϊ��Al3+���� |

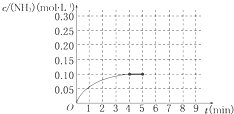 ��һ�ݻ�Ϊ2L���ܱ�������Ϊ���� 0.2mol��N2��0.6mol��H2����һ�������·������·�Ӧ��N2��g��+3H2��g��?2NH3��g����H��0��Ӧ��NH3�����ʵ���Ũ�ȵı仯�������ͼ��ʾ��
��һ�ݻ�Ϊ2L���ܱ�������Ϊ���� 0.2mol��N2��0.6mol��H2����һ�������·������·�Ӧ��N2��g��+3H2��g��?2NH3��g����H��0��Ӧ��NH3�����ʵ���Ũ�ȵı仯�������ͼ��ʾ��
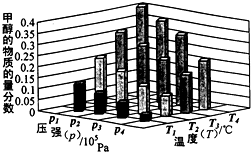 ��Դ����Լ���ҷ�չ���̵�����֮һ���״��������ѵȱ���Ϊ2 1���͵���ɫ��Դ����ҵ��������Ȼ��Ϊ��Ҫԭ���������̼��ˮ������һ���������Ʊ��ϳ�����CO��H2�������Ƴɼ״��������ѣ�
��Դ����Լ���ҷ�չ���̵�����֮һ���״��������ѵȱ���Ϊ2 1���͵���ɫ��Դ����ҵ��������Ȼ��Ϊ��Ҫԭ���������̼��ˮ������һ���������Ʊ��ϳ�����CO��H2�������Ƴɼ״��������ѣ�