��Ŀ����
�������ϰ�װ��Ч��ת��������ʹ����β���е���Ҫ��Ⱦ��(CO��NOx��̼�⻯����)�������Ӧ�����������ʣ���������β����Ⱦ��
(1)��֪��
N2(g)��O2(g)===2NO(g) ��H����180.5 kJ/mol��
2C(s)��O2(g)===2CO(g) ��H����221.0 kJ/mol��
C(s)��O2(g)===CO2(g) ��H����393.5 kJ/mol��
β��ת���ķ�Ӧ֮һ��2NO(g)��2CO(g)===N2(g)��2CO2(g)����H__________��
(2)ij�о���ѧϰС���ڼ�����Ա��ָ���£�����������̽��ij�ִ����ڲ�ͬ��ȼ��(������ȼ������������)�����¶�����β���Ĵ�Ч����
(1)��֪��
N2(g)��O2(g)===2NO(g) ��H����180.5 kJ/mol��
2C(s)��O2(g)===2CO(g) ��H����221.0 kJ/mol��
C(s)��O2(g)===CO2(g) ��H����393.5 kJ/mol��
β��ת���ķ�Ӧ֮һ��2NO(g)��2CO(g)===N2(g)��2CO2(g)����H__________��
(2)ij�о���ѧϰС���ڼ�����Ա��ָ���£�����������̽��ij�ִ����ڲ�ͬ��ȼ��(������ȼ������������)�����¶�����β���Ĵ�Ч����

��ʵ������г���ȼ�Ȳ�ͬ�⣬��������������β�������١�________�ȱ�����ͬ��
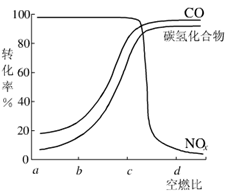
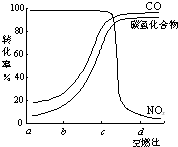
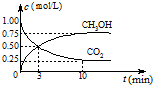
����һ�������£����β���е���Ҫ��Ⱦ���ת�������ȼ�ȵĹ�ϵ��ͼ��ʾ����ȼ��ԼΪ_______ʱ������������β���Ĵ�Ч����á�
����һ�������£����β���е���Ҫ��Ⱦ���ת�������ȼ�ȵĹ�ϵ��ͼ��ʾ����ȼ��ԼΪ_______ʱ������������β���Ĵ�Ч����á�
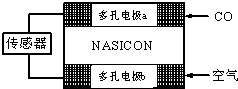
(3)CO��������ȼ�ϵ��Ϊ����ԭ������װ����ͼ��ʾ���õ���е����Ϊ�����ƣ������ƣ�����O2�������ڹ������NASICON�������ƶ�������˵���������___________��

A�������ĵ缫��ӦʽΪ��CO��O2����2e��===CO2
B������ʱ�缫b��������O2���ɵ缫a����缫b
C������ʱ�����ɵ缫aͨ������������缫b
D����������ͨ���ĵ���Խ��β����CO�ĺ���Խ��
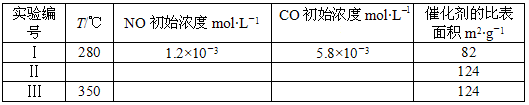
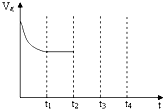
(4)�о���������ʹ�õ���������ʱ����������ȱ���������ѧ��Ӧ���ʡ�Ϊ�˷ֱ���֤�¶ȡ������ȱ�����Ի�ѧ��Ӧ���ʵ�Ӱ����ɣ�ijͬѧ���������ʵ�飬����ʵ�������Ѿ���������ʵ����Ʊ��У����ڱ���������ʣ���ʵ���������ݡ�
B������ʱ�缫b��������O2���ɵ缫a����缫b
C������ʱ�����ɵ缫aͨ������������缫b
D����������ͨ���ĵ���Խ��β����CO�ĺ���Խ��
(4)�о���������ʹ�õ���������ʱ����������ȱ���������ѧ��Ӧ���ʡ�Ϊ�˷ֱ���֤�¶ȡ������ȱ�����Ի�ѧ��Ӧ���ʵ�Ӱ����ɣ�ijͬѧ���������ʵ�飬����ʵ�������Ѿ���������ʵ����Ʊ��У����ڱ���������ʣ���ʵ���������ݡ�
(1)��746.5 kJ/mol
(2) �ٴ���Ӧ���¶ȣ���c
(3) B
(4)
(2) �ٴ���Ӧ���¶ȣ���c
(3) B
(4)


��ϰ��ϵ�д�
�����Ŀ

 CO2��SO2��NOx�ǶԻ���Ӱ��ϴ�����壬���ƺ�����CO2��SO2��NOx�ǽ������ЧӦ����������⻯ѧ��������Ч;����
CO2��SO2��NOx�ǶԻ���Ӱ��ϴ�����壬���ƺ�����CO2��SO2��NOx�ǽ������ЧӦ����������⻯ѧ��������Ч;����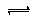 CO+3H2
CO+3H2
 Ŀǰ��ȫ���������Ϊ�����������������������������������Ҫԭ��֮һ���������������ж��ַ������������ϰ�װ��Ч��ת��������ʹ����β���е���Ҫ��Ⱦ�CO��NOx��̼�⻯����������Ӧ�����������ʣ���������β����Ⱦ��
Ŀǰ��ȫ���������Ϊ�����������������������������������Ҫԭ��֮һ���������������ж��ַ������������ϰ�װ��Ч��ת��������ʹ����β���е���Ҫ��Ⱦ�CO��NOx��̼�⻯����������Ӧ�����������ʣ���������β����Ⱦ��