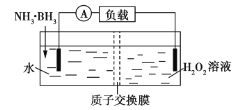
��Ŀ����
����Ŀ��ʵ�����Ʊ�������ʵ��װ�úͲ������£�
��ʵ�鲽�裩
ȡ100 mL�ձ�����20 mLŨ������18 mLŨ�������ƻ��Һ���������С�ļ���B�У���18 mL(15.84 g)������A�С��������µı�����μ�����ᣬ�ߵα߽��裬��Ͼ��ȣ���50��60���·�����Ӧ��ֱ����Ӧ������
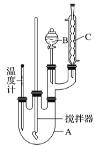
����ӦҺ��ȴ�����º����Һ©���У�����������ˮ��5% NaOH��Һ��ˮϴ�ӡ��ֳ��IJ��������ˮCaCl2����������Ƭ�̣���ȥCaCl2�������������ռ�205��210 ����֣��õ����������18.00 g����ش�
��1��װ��B��������________��װ��C��������________________��
��2��ʵ�����Ʊ��������Ļ�ѧ����ʽ��____________________________��
��3�����ƻ��Һʱ��_____����ܡ����ܡ�����Ũ������뵽Ũ�����У�˵������_________��
��4��Ϊ��ʹ��Ӧ��50��60 ���½��У����õķ�����__________��
��5����ϴ�Ӳ����У��ڶ���ˮϴ��������____________________��
���𰸡���Һ©�� 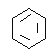 +HNO3
+HNO3![]()
 +H2O ����������ƽ����ѹ ���� �������Ž� ˮԡ���� ϴȥ������NaOH�����ɵ���
+H2O ����������ƽ����ѹ ���� �������Ž� ˮԡ���� ϴȥ������NaOH�����ɵ���
��������
(1)�������ṹ��������֪��,װ��BΪ��Һ©��,װ��CΪ����������,����Ũ���ᶼ�Իӷ�,����C��������������,Ŀ�����ԭ��������,
��ˣ�������ȷ����:��Һ©��;��������ƽ����ѹ;
��2��ʵ�����ñ���Ũ������ Ũ�������������������Ʊ����������仯ѧ��Ӧ����ʽΪ�� +HNO3
+HNO3![]()
 +H2O�����Ա���𰸣�
+H2O�����Ա���𰸣� +HNO3
+HNO3![]()
 +H2O��
+H2O��
(3)Ũ������Ũ�����ϻ�ų���������,�罫Ũ�������Ũ������,������ܶ�С��Ũ����,����Ϊ����Һ��Ž�,��˲��ܽ�Ũ������뵽Ũ�����У�
��ˣ��������:����,�������Ž�;
(4)��Ӧ��50��60 ���½���,����ˮ�ķе�,��������ˮԡ���ȿ���,���Ⱦ��ȣ�
��ˣ��������:ˮԡ����;
(5)����ˮϴ��ȥŨ���ᡢ����,�����������Ƴ�ȥ�ܽ��������,���ˮϴ��ȥδ��Ӧ��NaOH�����ɵ���,
��ˣ�������ǣ�ϴȥ������NaOH�����ɵ��Ρ�

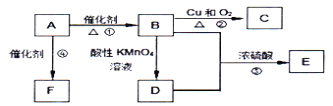
����Ŀ���������(K2FeO4)��һ���͡���Ч�����Ķ��ˮ��������
�����ϣ�K2FeO4Ϊ��ɫ���壬����KOH��Һ������ǿ�����ԣ������Ի�������Һ�п��ٲ���O2���ڼ�����Һ�н��ȶ���
��1���Ʊ�K2FeO4��

��AΪ��ȡ��������װ�ã�����ʢ��Ũ�������������Ϊ___________��
�ڳ���װ��B�е��Լ�Ϊ___________��
��CΪ�Ʊ�K2FeO4װ�ã�KOH��Һ������ԭ����___________��
��DΪβ������װ�ã�������Ӧ�����ӷ���ʽΪ___________��
��2��̽�� K2FeO4�����ʡ�ȡC����ɫ��Һ������ϡ���ᣬ�����S��ɫ���壬����Һa�������������к���Cl2��Ϊ֤��K2FeO4�ܷ�����Cl��������Cl2��������·�����
����I | ȡ������Һa���μ�KSCN��Һ����������Һ�ʺ�ɫ�� |
������ | ��KOH��Һ���ϴ��C�����ù��壬����KOH��Һ��K2FeO4�ܳ����õ���ɫ��Һb��ȡ����b���μ����ᣬ��Cl2������ |
���ɷ���I����Һ����֪a�к���___________�������ӵIJ���___________(������������������)�ж�һ������K2FeO4��Cl����ԭ���γɵġ�
�ڷ�������KOH��Һϴ�ӵ�Ŀ����___________���������ó������ԣ�Cl2___________FeO42��(����>������<��)
��3��ʹ��ʱ����ͨ���ⶨ������صĴ������ж����Ƿ���ʡ�K2FeO4��������Һ�з�Ӧ������_______ FeO42��+______H+===_______O2��+________Fe3++________(��ƽ������������ӷ���ʽ)________����ȡC��ϴ�Ӳ��������Ʒ������10g������ϡ���ᣬ�ռ���0.672L����(��״����)������Ʒ�и�����ص���������ԼΪ___________��(������������0.1%)