��Ŀ����
����˵����ȷ����(����)
��SiO2����HF��Ӧ����������ܱ����ڲ���ƿ��
��ʵ������ȡ����ʱ��Ϊ�˷�ֹ������Ⱦ���������������������������Һ����
�����ˮ����μ�����������FeCl3��Һ�����Ƶ�Fe(OH)3����
��Na2O��Na2O2���Ԫ����ͬ���������������Ӹ�����Ҳ��ͬ
�ݳ�ȥHC l�����е�Cl2���ɽ�����ͨ�뱥��ʳ��ˮ��
l�����е�Cl2���ɽ�����ͨ�뱥��ʳ��ˮ��
������ˮ�м�CaCO3��ĩ���������Һ��HClO��Ũ��
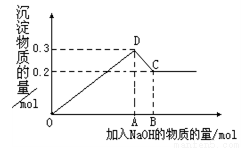
��SiO2����NaOH��Һ��HF��Һ��Ӧ������SiO2������������
A. 3�� B. 4�� C. 5�� D. 6��
��ϰ��ϵ�д�
 ��Уͨ��֤��Ч��ҵϵ�д�
��Уͨ��֤��Ч��ҵϵ�д�
�����Ŀ
�����������Һ��ϡ���ᷴӦ�Ļ�ѧ����ʽΪ��
Na2S2O3 + H2SO4 = Na2SO4 + SO2 + S�� + H2O
���и���ʵ�������ȳ��ֻ��ǵ���
ʵ�� | ��Ӧ�¶�/�� | Na2S2O3��Һ | ϡH2SO4 | H2O | ||
V/mL | c/(mol��L-1) | V/mL | c/(mol��) | V/mL | ||
A | 25 | 5 | 0.1 | 10 | 0.1 | 5 |
B | 25 | 5 | 0.2 | 5 | 0.2 | 10 |
C | 35 | 5 | 0.1 | 10 | 0.1 | 5 |
D | 35 | 5 | 0.2 | 5 | 0.2 | 10 |
A. A B. B C. C D. D


 CO(g)��3H2(g)���������ʵ�ȼ�����������±�����֪1 mol H2O(g)ת��Ϊ1 mol H2O(l)ʱ�ų�44.0 kJ������д��CH4��H2O�ڸ����·�Ӧ���Ȼ�ѧ����ʽ�� _______________________________��
CO(g)��3H2(g)���������ʵ�ȼ�����������±�����֪1 mol H2O(g)ת��Ϊ1 mol H2O(l)ʱ�ų�44.0 kJ������д��CH4��H2O�ڸ����·�Ӧ���Ȼ�ѧ����ʽ�� _______________________________��