��Ŀ����
Ϊ�ⶨijƷ��ϴ������(����)������ijѧ��������֪���ʵ���Ũ�ȵ�NaOH��Һ���ⶨ��ϴ��(����)�����ʵ���Ũ��ʱ��ѡ���̪��ָʾ��������д���пհף�
��1����ʽ�ζ��ܵ�ʹ�÷�������ȷ������Ⱥ�˳��Ϊ����ѡ����ţ���
A��������������Һ��ϴ��������������������Һ
B���ų�����������Һ���еζ�
C��������ʼ����
D����©����ˮϴ2-3��
��2���ñ���NaOH��Һ�ζ����������ʱ,���ֿ��Ƽ�ʽ�ζ��ܵIJ���������ҡ����ƿ���۾�Ӧע�� ��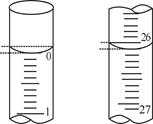
��3�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ������ͼ��ʾ��������NaOH��Һ�����Ϊ mL��
��4��ijѧ����������ʵ��ֱ��¼�й��������±���
| �ζ����� | ������������/mL | 0.1000 mol/LNaOH��Һ�����/mL | ||
| �ζ�ǰ�̶� | �ζ���̶� | ��Һ���/mL | ||
| ��һ�� | 25.00 | 2.00 | 28.15 | 26.15 |
| �ڶ��� | 25.00 | 1.50 | 29.50 | 28.00 |
| ������ | 25.00 | 0.20 | 26.55 | 26.35 |
��ѡ�����к��������ݼ����ϴ������(����)�����ʵ���Ũ�ȣ�
c(HCl)�� ��
��5���������в���������ϴ������(����)��Ũ�ȵ�Ӱ�죺���ƫ����ƫС�� ����Ӱ�족��
A����ȡ����Һʱ����ʼ���Ӷ����� ���Ӷ������� ��
B������ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ���� ��
(1) DACB��
��2����ƿ����Һ��ɫ�ı仯��
��3��26.10mL��
��4�� c(HCl)��0.105 mol/L��
��5��ƫ��ƫ��
���������������1���������裺�����������ϴ������װҺ��������ʼ�������к͵ζ����յ��жϡ����㡢���ݴ�����
��2���ζ�ʱ�����ֿ��ƻ��������ְ�˳ʱ��ҡ����ƿ���۾��۲���ƿ����Һ��ɫ�ı仯��
��3���۲����ʱҪƽ�ӣ�
��4��c(HCl)��0.1000*��26.35+26.15��/2/25.00="0.105" mol/L��
��5��ȡ����Һʱ�����Ӻ��ӣ���ʹ����Һ�Ķ���ƫС ��ʹ����ϴ������(����)��Ũ��ƫ�����ζ����������ݣ���ʹ��Һ�������ƫ�Ӷ�ʹ����ϴ������(����)��Ũ��ƫ��
���㣺�������顣

A��B��C��D������������ˮ����ȫ���룬��������������±���
| ������ | Na+��Al3+��Ba2+��H+��NH4+ |
| ������ | SO42-��OH-��CO32-��Cl- |
�ֽ�������ʵ�飺
������A��Һ��B��Һ��Ϲ��ȿ����ɳ����ʹ̼�����ζ���壻
������A��Һ��C��Һ��Ͽ����ɳ����ң�
��A��Һ��B��Һ�����ܽ�����ң����������ܽ�����ס�
��ش�
��1��A�Ļ�ѧʽΪ_________������ʱ����pH��ȵ�A��Һ��D��Һ�ֱ�ϡ��10����pH�ֱ��Ϊa��b����a _______b(�>������=����<������
��2����������C��Һ�����գ�������ù���Ϊ_______ (�ѧʽ����
��3��C��Һ��D��Һ��Ӧ�����ӷ���ʽΪ_______
��4����B��Һ����μ��������������ʵ���Ũ�ȵ�NaOH��Һ���μӹ�����ˮ�ĵ���ƽ�⽫_______ (������������������ƶ�������������Һ�и�����Ũ���ɴ�С��˳��Ϊ____________________
��5����֪������Ksp=x����0.03mol��L-1��A��Һ��0.01mol��L-1��B��Һ�������ϣ������Һ��������ӵ�Ũ��Ϊ_______ (�ú�x�Ĵ���ʽ��ʾ����Ϻ���Һ����仯���Բ��ƣ���
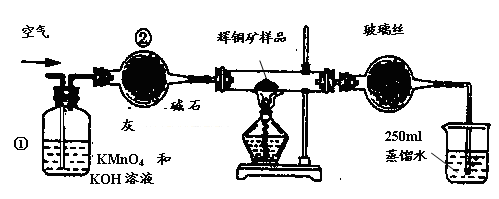
��ҵ��Ϊ�˲ⶨ��ͭ����Ҫ�ɷ���Cu2S����Cu2S�������������������ͼװ�á�ʵ��ʱ�����²��������
| A������ȫ��������ʹ���Ϊ��ͼװ�ã������װ�õ������ԡ� |
| B����ȡ��ϸ�Ļ�ͭ����Ʒ1.000g�� |
| C���������õ���ƷС�ĵط���Ӳ�ʲ������С� |
| D����ÿ����1L�����ʹ�������� |
F. ��ȡ25.00ml��SO2��ˮ��Һ��250ml��ƿ�У���0.0100mol/L KMnO4����Һ�ζ����յ㡣���������������ظ��ζ�2��3�Ρ�
�Իش��������⣺
��1��װ�âٵ�������_________________��װ�âڵ�������____________________��
��2���ٶ���ͭ���е���ȫ��ת��ΪSO2������ȫ����ˮ���գ������F����������Ӧ�Ļ�ѧ����ʽΪ ��������_______________________________������ʱ���жϵζ��Ѿ��ﵽ�յ㡣
��3��������F�ĵζ�������±���ʾ�����ͭ����Ʒ��Cu2S������������_________��
| �ζ� ���� | ������Һ�� ���/mL | ����Һ����� | |
| �ζ�ǰ�̶�/mL | �ζ���̶�/mL | ||
| 1 | 25.00 | 1.04 | 21.03 |
| 2 | 25.00 | 1.98 | 21.99 |
| 3 | 25.00 | 3.20 | 21.24 |
��4���������������һ�����Ե�ȱ��Ӱ���˲ⶨ����������ڲ���ʧ������Ϊ�� ��дһ�ּȿɣ���
��5����֪�ڳ�����FeS �� Ksp�� 6 . 25 �� 10 ��18, H2S ������Һ�� c (H������ c (S2����֮��������¹�ϵ�� c2 (H��) ��c��S2��) =" 1" . 0��10��22���ڸ��¶��£������� FeS Ͷ�����ⱥ����Һ�У���ʹ��Һ��c��Fe2+��Ϊ lmol/L��Ӧ������Һ��c��Hʮ��Ϊ__________������
ij�о���ѧϰС��Ϊ��̽������ĵ������������������ʵ�顣
ʵ��һ�����Ʋ��궨������Һ��Ũ��
ȡ����������250 mL 0.2 mol��L��1�Ĵ�����Һ����0.2 mol��L��1�Ĵ�����Һϡ�ͳ�����Ũ�ȵ���Һ������NaOH����Һ�����������Һ��Ũ�Ƚ��б궨���ش��������⣺
(1)����250 mL 0.2 mol��L��1������Һʱ��Ҫ�õ��IJ�����������Ͳ���ձ����������� �� ��
(2)Ϊ�궨ij������Һ��ȷŨ�ȣ���0.200 0 mol��L��1��NaOH��Һ��20.00 mL������Һ���еζ������εζ�����NaOH��Һ��������£�
| ʵ����� | 1 | 2 | 3 | 4 |
| ����NaOH��Һ�����(mL) | 20.05 | 20.00 | 18.80 | 19.95 |
��ô�����Һ��ȷŨ��Ϊ (����С�������λ)��
ʵ�����̽��Ũ�ȶԴ������̶ȵ�Ӱ��
��pH�Ʋⶨ25��ʱ��ͬŨ�ȵĴ����pH��������£�
| ����Ũ��(mol��L��1) | 0.001 0 | 0.010 0 | 0.020 0 | 0.100 0 | 0.200 0 |
| pH | 3.88 | 3.38 | 3.23 | 2.88 | 2.73 |
�ش��������⣺
(1)���ݱ������ݣ����Եó�������������ʵĽ��ۣ�����Ϊ�ó��˽��۵������ǣ� ��
(2)�ӱ��е����ݣ������Եó���һ���ۣ����Ŵ���Ũ�ȵļ�С������ĵ���̶Ƚ� (�������С�����䡱)��
ʵ������̽���¶ȶԴ������̶ȵ�Ӱ��
�������һ��ʵ����ɸ�̽������������ʵ�鷽����
��
ʵ������NaOH��������0.1 000mol��L-1NaOH��Һ500mL��
��1����������ƽ��ȡNaOH����________g����Һ���ƹ����õ����в������������״�ʹ�õ��Ⱥ�˳��������________ (������ѡ�����)
A�������� B����ͷ�ι� C���ձ� D��500mL����ƿ
��2���������Ƶ�0.1000mol��L-1NaOH��Һͨ���к͵ζ��ⶨһԪ����HA��ҺŨ�ȣ�ÿ�εζ�ȡ�õ�HA��Һ��Ϊ20.00mL��ʹ�÷�̪��ҺΪָʾ�����ζ��յ�ı�־��____________________________���ζ���ʵ�����ݼ�¼��
| �ζ����� | NaOH��Һ�����mL�� | |
| V1 | V2 | |
| 1 | 3.05 | 44 |
| 2 | 1.45 | 41.5 |
| 3 | 7.65 | 47.6 |
��������ʵ�����ݣ���ø�HA��Һ���ʵ���Ũ��Ϊ____________________��
��3�������к͵ζ�ʵ���У����²������ܵ���������ҺŨ��ƫ�ߵ���__________(��ѡ�����)
A���ζ����ô�װҺ��ϴ
B����ƿ�ô�װҺ��ϴ
C���ζ�ǰ�ζ���ĩ������û�Ͼ�
D���ζ�ǰƽ�ӹ۲�������ζ����ӹ۲����
E�����õı�Һ(NaOH��Һ)Ũ��ƫ��
��4����pH�Ʋ�ø�HA��ҺpH=a����������к͵ζ����������ʵ���¶���HA��Ka=________��
ijѧ������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�NaOH��Һ��
������ɷֽ�Ϊ���¼�����
a. ��ȡ20.00mL�����NaOH��Һע��ྻ����ƿ��������2-3�η�̪
b. �ñ�������Һ��ϴ�ζ���2-3��
c. ��ʢ�б���Һ����ʽ�ζ��̶ܹ��ã�����Һ��ʹ�ζ��ܼ��������Һ
d. ȡ��������Һע����ʽ�ζ�����0�̶�����2-3cm
e. ����Һ����0��0�̶����£����¶���
f. ����ƿ���ڵζ��ܵ����棬�ñ�������Һ�ζ����յ㣬���µζ���Һ��Ŀ̶�
���������գ�
��1����ȷ������˳���ǣ��������ĸ��д��__________________ ____��
��2���ζ��յ�ʱ��Һ����ɫ�仯�� ��
��3�����в����п���ʹ����NaOH��Һ��Ũ����ֵƫ�͵���_____ ___��
| A����ʽ�ζ���δ�ñ�������ϴ��ֱ��ע������� |
| B���ζ�ǰʢ��NaOH��Һ����ƿ������ˮϴ����û�и��� |
| C����ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ |
| D����ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ��� |
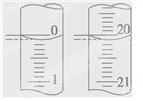
��5��ijѧ������3��ʵ��ֱ��¼�й��������±���
| �ζ� ���� | ����NaOH��Һ�����/mL | 0.1000mol/L��������/mL | ||
| �ζ�ǰ�̶� | �ζ���̶� | ��Һ���/mL | ||
| ��һ�� | 25.00 | 0.20 | 20.22 | |
| �ڶ��� | 25.00 | 0.56 | 24.54 | |
| ������ | 25.00 | 0.42 | 20.40 | |
�����ϱ��������NaOH��Һ�����ʵ���Ũ��Ϊ ��
ijԭ���װ������ͼ��ʾ������ܷ�ӦΪ2Ag��Cl2��2AgCl������˵����ȷ����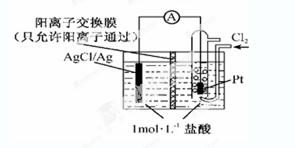
| A��������ӦΪAgCl ��e����Ag ��Cl�� |
| B���ŵ�ʱ������Ĥ�Ҳ���Һ���д�����ɫ�������� |
| C������NaCl��Һ�������ᣬ�����ܷ�Ӧ��֮�ı� |
| D������·��ת��0.01 mol e��ʱ������Ĥ�����Һ��Լ����0.02 mol���� |
�������ϲ����������ԭ��صĻ�ѧ��Ӧ��
| A��2FeCl3(aq)��2KI(aq) = 2FeCl2(aq)��2KCl(aq)��I2(aq) ��H <0 |
| B��Ba(OH)2��8H2O(s)+2NH4Cl(s) = BaCl2(aq)+2NH3��H2O(aq)+8H2O��1�� ��H >0 |
| C��4Al(s)+ 6H2O��1��+ 3O2(g)==4Al(OH)3(s) ��H <0 |
| D��Zn(s)+2MnO2(s)+2H2O��1�� = 2MnOOH(s) +Zn(OH)2(s) ��H <0 |
