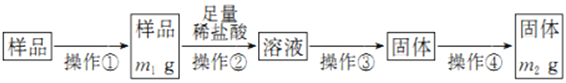
��Ŀ����
����Ŀ��A��B��C�ǵ��ʣ�����A�ǽ������������ʼ��ת����ϵ��ͼ��
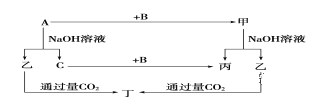
����ͼʾת����ϵ�ش�
(1)д���������ʵĻ�ѧʽ��A_______��B___________����__________����_______��
(2)д�����б仯�ķ���ʽ��
��A��NaOH��Һ��Ӧ�Ļ�ѧ����ʽ_______________________��
�ڼ���NaOH��Һ��Ӧ�����Һͱ������ӷ���ʽ_______________________��
(3)��һ������A���뵽NaOH��Һ�У�������C�ڱ�״���µ����Ϊ3.36 L�������ĵ�A�����ʵ���Ϊ________��ת�Ƶ��ӵ����ʵ���Ϊ________��
���𰸡�Al O2�� NaAlO2 Al(OH)3 2Al��2NaOH��2H2O===2NaAlO2��3H2�� Al2O3��2OH��===2AlO2����H2O 0.1 mol 0.3 mol
��������
A�ǽ������ʣ�A����NaOH��Һ��Ӧ��������������NaOH��Һ��Ӧ��ΪAl����AΪAl��Al��NaOH��Һ��Ӧ����NaAlO2��H2��C�ǵ��ʣ�CΪH2����ΪNaAlO2��B�ǵ��ʣ�Al+B���ף���+NaOH����+NaAlO2��H2+B��������BΪO2����ΪAl2O3����ΪH2O����ƫ��������Һ��ͨ�������̼����������������������Al(OH)3���ݴ��жϡ�
�������Ϸ�����֪AΪAl��BΪO2��CΪH2����ΪAl2O3����ΪNaAlO2����ΪH2O������Al(OH)3����
��1��A�Ļ�ѧʽΪAl��B�Ļ�ѧʽΪO2���ҵĻ�ѧʽΪNaAlO2�����Ļ�ѧʽΪAl(OH)3��
��2����Al��NaOH��Һ��Ӧ����ƫ�����ƺ���������Ӧ�Ļ�ѧ����ʽΪ2Al+2NaOH+2H2O��2NaAlO2+3H2����
��Al2O3��NaOH��Һ��Ӧ����ƫ�����ƺ�ˮ����Ӧ�����ӷ���ʽΪAl2O3��2OH����2AlO2����H2O��
��3��n��H2��=3.36L��22.4L/mol=0.15mol�����ݷ�Ӧ2Al+2NaOH+2H2O��2NaAlO2+3H2����֪n��Al��=2/3��0.15mol=0.1mol����Ӧ����1molAlת��3mol���ӣ���÷�Ӧ��ת�Ƶ������ʵ���Ϊ0.3mol��

 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д�