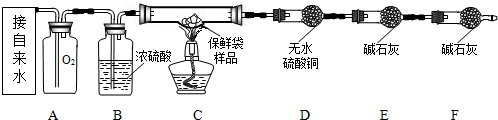
��Ŀ����
ijͬѧ��ʵ����⣺��ȥͭ���л��е�����ͭ��ĩ�����������ʵ�鷽��������������ԭ����ͭ ����һ����̼��ԭ����ͭ ����̼�ۻ�ԭ����ͭ ����ϡ�����ܽ�����ͭ��CuO+H2SO4=CuSO4+H2O������������һͬѧ�Ը�ͬѧ��Ʒ��������ۣ�����Ϊ˵�����������ǣ�������
������A����������ԭ����ͭ����ͭ��ˮ�����������Ⱦ��
B����ϡ�����ܽ�����ͭ��CuO+H2SO4=CuSO4+H2O����ͭ�������ᷴӦ��
C����һ����̼��ԭ����ͭ��һ����̼�ж��������β�����գ������鷳��
D����̼�ۻ�ԭ����ͭ��̼�ǹ��壬�������ÿ��ƣ����״���ϡ�����ʣ�
B����ϡ�����ܽ�����ͭ��CuO+H2SO4=CuSO4+H2O����ͭ�������ᷴӦ��
C����һ����̼��ԭ����ͭ��һ����̼�ж��������β�����գ������鷳��
D����̼�ۻ�ԭ����ͭ��̼�ǹ��壬�������ÿ��ƣ����״���ϡ�����ʣ�
����⣺A����������ԭ����ͭ����ͭ��ˮ�����������Ⱦ����A����
B����ϡ�����ܽ�����ͭ��CuO+H2SO4=CuSO4+H2O����ͭ�������ᷴӦ������������У�Ҳ����Ҫ���ȣ���B��ȷ��
C����һ����̼��ԭ����ͭ��һ����̼�ж��������β�����գ������鷳����C��ȷ��
D����̼�ۻ�ԭ����ͭ��̼�ǹ��壬�������ÿ��ƣ����״���ϡ�����ʣ���D��ȷ��
��ѡ��A��
B����ϡ�����ܽ�����ͭ��CuO+H2SO4=CuSO4+H2O����ͭ�������ᷴӦ������������У�Ҳ����Ҫ���ȣ���B��ȷ��
C����һ����̼��ԭ����ͭ��һ����̼�ж��������β�����գ������鷳����C��ȷ��
D����̼�ۻ�ԭ����ͭ��̼�ǹ��壬�������ÿ��ƣ����״���ϡ�����ʣ���D��ȷ��
��ѡ��A��
������Ҫ��ȥ������е����ʣ�ͨ��������˼·���ٽ����ʴӻ�����г�ȥ���ڽ��������ʴӻ������ȡ����

��ϰ��ϵ�д�
 �����ƻ���ĩ��̶�100��ϵ�д�
�����ƻ���ĩ��̶�100��ϵ�д� �ܿ���ȫ��100��ϵ�д�
�ܿ���ȫ��100��ϵ�д�
�����Ŀ