��Ŀ����
 ˮ��������ԴȪ��Ϊ���������Ŀɳ�����չ������Ӧ���˽�ˮ������ˮ��Դ��
ˮ��������ԴȪ��Ϊ���������Ŀɳ�����չ������Ӧ���˽�ˮ������ˮ��Դ����1����Ȼˮ�к����������ʣ�����ˮ��ͨ������Ȼˮ���г�����
����
����
�������������ȴ�����ʹ֮�ﵽ����ˮ������ʵ�������Ҫ�õ������̶���ߵ�ˮ����������
����
�ķ�������2������Ӳ�ȹ����ˮ���������彡��������ˮ��Ӳˮ������ˮ�����õ�������
����ˮ
����ˮ
��������ʹӲˮ�����ij��÷��������
���
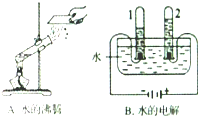
����3������ͼ��ʾ������ʵ���У����������仯����ʵ��
ˮ�ķ���
ˮ�ķ���
��ʵ��B��û�з����ı����������ԭ�Ӻ���ԭ��
��ԭ�Ӻ���ԭ��
���Թ�2�еõ�������������
����
����4����Լ��ˮ��ÿ���������������Ӧ���ᳫ���·�ʽ�е�
AD
AD
������ĸ��ţ���A��������ˮ��ϴ��ˮ����������� B������ϵط�ˮϴ�·�
C������ϵط�ˮˢ�� D������ࡢ�ι�ķ�������ũ����֣�
��������1�����ݾ�ˮ�ķ����Լ������̶ȵķ�����������н��
��2�������÷���ˮ����Ӳˮ����ˮ�������г�����з���ʹӲˮ�������н��
��3������ˮ���ȷ���û���������������������仯����ˮͨ��ֽ�����������������ԭ���ǻ�ѧ�仯����С�����������������������Ϊ2��1���н��
��4������ֻҪ���Ͻ�Լ��ˮ���н��н��
��2�������÷���ˮ����Ӳˮ����ˮ�������г�����з���ʹӲˮ�������н��
��3������ˮ���ȷ���û���������������������仯����ˮͨ��ֽ�����������������ԭ���ǻ�ѧ�仯����С�����������������������Ϊ2��1���н��
��4������ֻҪ���Ͻ�Լ��ˮ���н��н��
����⣺��1����Ȼˮ�к����������ʣ�����ˮ��ͨ������Ȼˮ���г��������ˡ�������������ʹ֮�ﵽ����ˮ������ʵ�������Ҫ�õ������̶���ߵ�ˮ����������ķ�����������ˣ�����
��2���÷���ˮ����Ӳˮ����ˮ��������ĭ�������ˮ����ĭ�ٵ���Ӳˮ�������г�����з���ʹӲˮ�������������ˮ����У�
��3��ˮ���ȷ���û���������������������仯����ˮͨ��ֽ��������������������иù�������ԭ�Ӻ���ԭ��û�иı䣬�����������������Ϊ2��1�������Թ�2�еõ������������������ˮ�ķ��ڣ���ԭ�Ӻ���ԭ�ӣ�������
��4��A��������ˮ��ϴ��ˮ���������������������һˮ���ã����Խ�Լ��ˮ��
B������ϵط�ˮϴ�·��������˷�ˮ��Դ��
C������ϵط�ˮˢ���������˷�ˮ��Դ��
D���ִ�ũ����ֲ�����ࡢ�ι�ȷ�ʽ�����Խ�Լ��ˮ����
��ѡ��AD��
��2���÷���ˮ����Ӳˮ����ˮ��������ĭ�������ˮ����ĭ�ٵ���Ӳˮ�������г�����з���ʹӲˮ�������������ˮ����У�
��3��ˮ���ȷ���û���������������������仯����ˮͨ��ֽ��������������������иù�������ԭ�Ӻ���ԭ��û�иı䣬�����������������Ϊ2��1�������Թ�2�еõ������������������ˮ�ķ��ڣ���ԭ�Ӻ���ԭ�ӣ�������
��4��A��������ˮ��ϴ��ˮ���������������������һˮ���ã����Խ�Լ��ˮ��
B������ϵط�ˮϴ�·��������˷�ˮ��Դ��
C������ϵط�ˮˢ���������˷�ˮ��Դ��
D���ִ�ũ����ֲ�����ࡢ�ι�ȷ�ʽ�����Խ�Լ��ˮ����
��ѡ��AD��
�����������ؼ���Ҫ֪������ˮ�ľ������̣�֪�������г�����Լ��ˮ�ķ�����

��ϰ��ϵ�д�
�����Ŀ
 ˮ��������ԴȪ��Ϊ���������Ŀɳ�����չ������Ӧ���˽�ˮ���й�֪ʶ��С��ͬѧ������һ������ˮ������ͼ��ʾ��
ˮ��������ԴȪ��Ϊ���������Ŀɳ�����չ������Ӧ���˽�ˮ���й�֪ʶ��С��ͬѧ������һ������ˮ������ͼ��ʾ��
 ˮ��������ԴȪ��Ϊ���������Ŀɳ�����չ������Ӧ���˽�ˮ���й�֪ʶ��
ˮ��������ԴȪ��Ϊ���������Ŀɳ�����չ������Ӧ���˽�ˮ���й�֪ʶ��