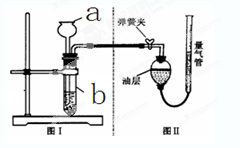
��Ŀ����
������ij��ȤС��ͬѧ����3��Сʵ�飮����ʵ�����ݻش��������⣨�����Ĺ̶�װ���Ѿ�ʡ�ԣ���
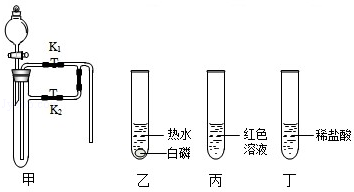
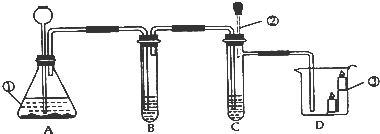
��1��������Ϊ�������̣����Ӽ���װ�ã��� ������ء�K1���͡�K2���IJ��������ӷ�Һ©������м�������˫��ˮ����ʵ�����֤����ȼ��ȼ����Ҫ�������Ӵ������з�����Ӧ�Ļ�ѧ����ʽΪ�� ����
��2��������Ϊˮ������Ϊ��ɫ��Һ�����Ӽױ�����K1���ر�K2���ӷ�Һ©������м���ijҩƷ����ֻ������Һ�ɺ�ɫ��Ϊ��ɫ��д�����з�����Ӧ�Ļ�ѧ����ʽ�� ����
��3��������Ϊ������̼��K1��K2�رգ������Ӽ����ӷ�Һ©������м��������ij���ʯ��ˮ��һ��ʱ���K1������ʵ������м��й۲쵽�������� ����

��1��������Ϊ�������̣����Ӽ���װ�ã��� ������ء�K1���͡�K2���IJ��������ӷ�Һ©������м�������˫��ˮ����ʵ�����֤����ȼ��ȼ����Ҫ�������Ӵ������з�����Ӧ�Ļ�ѧ����ʽΪ�� ����
��2��������Ϊˮ������Ϊ��ɫ��Һ�����Ӽױ�����K1���ر�K2���ӷ�Һ©������м���ijҩƷ����ֻ������Һ�ɺ�ɫ��Ϊ��ɫ��д�����з�����Ӧ�Ļ�ѧ����ʽ�� ����
��3��������Ϊ������̼��K1��K2�رգ������Ӽ����ӷ�Һ©������м��������ij���ʯ��ˮ��һ��ʱ���K1������ʵ������м��й۲쵽�������� ����
��1���ر�K1����K2��2H2O2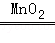 2H2O+O2����2��2NaOH+H2SO4=Na2SO4+2H2O
2H2O+O2����2��2NaOH+H2SO4=Na2SO4+2H2O
��3������ʯ��ˮ����ǣ���K1����Һ�嵹����ף�������ɫ���ݣ����Ǽ��٣���ʧ��������Һ�����붡��
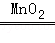 2H2O+O2����2��2NaOH+H2SO4=Na2SO4+2H2O
2H2O+O2����2��2NaOH+H2SO4=Na2SO4+2H2O��3������ʯ��ˮ����ǣ���K1����Һ�嵹����ף�������ɫ���ݣ����Ǽ��٣���ʧ��������Һ�����붡��
�����������1������Һ���Ὣ�����Թܵײ��ĵ��ܿ�û��Һ���ڣ��������ų������Թر�K1����K2ʹ�����K2�ĵ����ų�����Ӧ���ǹ������⣬��������ˮ���������ù۲취��ƽ������������������д�ڵȺŵ��ϱߣ����Է���ʽ�ǣ�2H2O2
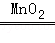 2H2O+O2������2�����з�̪������������Һ�Ժ�ɫ�������ᷢ����Ӧ���Ա����������������Ʒ�Ӧ��ͱ����ɫ�����Դӷ�Һ©������м���Ũ���ᣬŨ��������ˮ�ų��������ȣ�ʹ�Թ���ѹǿ��������ѹ����Թ��ڣ��������������Ʒ�����Ӧ��ʹ��ɫ��ȥ�����Է�Ӧ�����������ƺ�����д�ڵȺŵ���ߣ��������������ƺ�ˮ��д�ڵȺŵ��ұߣ��ù۲취��ƽ�����Է���ʽ�ǣ�2NaOH+H2SO4=Na2SO4+2H2O����3���ӷ�Һ©������м��������ij���ʯ��ˮ����Ϊ������̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ�����Ա���ǣ�������̼���ٺ��Թ���ѹǿ��С����K1�����ڴ���ѹ�������£���ѹ���Թ��ڣ�������̼��Ʒ�Ӧ����������̼�����Ǽ��ٻ���ʧ���������ݲ�������������ѹǿ�����ֽ�����Һ��ѹ�붡�У�
2H2O+O2������2�����з�̪������������Һ�Ժ�ɫ�������ᷢ����Ӧ���Ա����������������Ʒ�Ӧ��ͱ����ɫ�����Դӷ�Һ©������м���Ũ���ᣬŨ��������ˮ�ų��������ȣ�ʹ�Թ���ѹǿ��������ѹ����Թ��ڣ��������������Ʒ�����Ӧ��ʹ��ɫ��ȥ�����Է�Ӧ�����������ƺ�����д�ڵȺŵ���ߣ��������������ƺ�ˮ��д�ڵȺŵ��ұߣ��ù۲취��ƽ�����Է���ʽ�ǣ�2NaOH+H2SO4=Na2SO4+2H2O����3���ӷ�Һ©������м��������ij���ʯ��ˮ����Ϊ������̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ�����Ա���ǣ�������̼���ٺ��Թ���ѹǿ��С����K1�����ڴ���ѹ�������£���ѹ���Թ��ڣ�������̼��Ʒ�Ӧ����������̼�����Ǽ��ٻ���ʧ���������ݲ�������������ѹǿ�����ֽ�����Һ��ѹ�붡�У�
��ϰ��ϵ�д�
�����Ŀ
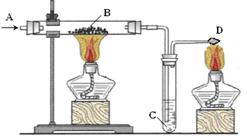
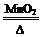 2KCl+3O2����
2KCl+3O2����