��Ŀ����
��ͼ��ʾΪʵ�����г����������Ʊ����ռ�װ�á���ش��������⣺
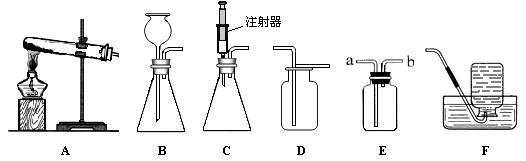
��1��ʵ�����ø��������ȡ������Ӧѡ�÷���װ�� �� (����ĸ���)����Ӧ�Ļ�ѧ����ʽΪ �� ������װ��D�ռ������������ķ����� �� ��
��2��ʵ�����ù���������Һ�Ͷ���������ȡ��������ѡ��C������װ�ã�����Ϊѡ��C���ŵ��� ��
��3����ʹ��װ��E���ſ������ռ�������̼��������Ӧ�� �� ���a����b������ͨ�룻��ʹ��װ��E����ˮ���ռ��������Ƚ�ƿ��װ��ˮ���ٽ������ �� ���a����b������ͨ�롣
��4����֪һ����������������ˮ���ڿ���������������������Ӧ�����ռ�һ����������ʱӦѡ��ͼ��װ�� (����ĸ���)��

��1��ʵ�����ø��������ȡ������Ӧѡ�÷���װ�� �� (����ĸ���)����Ӧ�Ļ�ѧ����ʽΪ �� ������װ��D�ռ������������ķ����� �� ��
��2��ʵ�����ù���������Һ�Ͷ���������ȡ��������ѡ��C������װ�ã�����Ϊѡ��C���ŵ��� ��
��3����ʹ��װ��E���ſ������ռ�������̼��������Ӧ�� �� ���a����b������ͨ�룻��ʹ��װ��E����ˮ���ռ��������Ƚ�ƿ��װ��ˮ���ٽ������ �� ���a����b������ͨ�롣
��4����֪һ����������������ˮ���ڿ���������������������Ӧ�����ռ�һ����������ʱӦѡ��ͼ��װ�� (����ĸ���)��
��1��A 2KMnO4 K2MnO4 + MnO2+ O2��
K2MnO4 + MnO2+ O2��
�������ǵ�ľ�����ڼ���ƿ�ڣ�ľ����ȼ��֤���Ѽ���
��2���������������Ŀ��������Ķ���
��3��a b ��4��F
 K2MnO4 + MnO2+ O2��
K2MnO4 + MnO2+ O2���������ǵ�ľ�����ڼ���ƿ�ڣ�ľ����ȼ��֤���Ѽ���
��2���������������Ŀ��������Ķ���
��3��a b ��4��F
�����������1����������ڼ��ȵ������£��ֽ���������ء��������̺��������ʷ�Ӧ�Ļ�ѧ����ʽΪ2KMnO4��K2MnO4+MnO2+O2�������ڸ÷�Ӧ�ǹ����ڼ��������·ֽ⣬��Ӧѡ�÷���װ��A������������ʹ�����ǵ�ľ����ȼ���ʿɽ������ǵ�ľ�����ڼ���ƿ�ڣ���ľ����ȼ����֤���Ѽ�����
��2������Cװ�����õ���ע���������ӹ���������Һ��������ͨ�����Ʒ�Ӧ����������Һ���������������������Ŀ��������Ķ��١�
��3�����ڶ�����̼���ܶȱȿ��������������ſ������ռ�������װ��E���С��ſ��������ռ�������̼���������̼Ӧ�ӽϳ��ĵ���a���룬���������Ͻ������ӽ϶̵ĵ����ų�������װ��E����ˮ���ռ��������������������ܶ�С��ˮ�����Ҫ�ӽ϶̵ĵ���bͨ�룬���������½�ˮ�ӳ����ų���
��4���������⣬����һ����������������ˮ�����ڿ���������������������Ӧ�����ռ�һ����������Ҫ����ˮ���ռ�����ѡF
���������������ʵ�����Ʒ���ԭ����ע������ȣ��ǽ���Ĺؼ������⣬������ص��dz�������ķ���װ�ú��ռ�װ�õ�ѡȡ������Ҫѧ����ݷ�Ӧ���״̬�ͷ�Ӧ������ѡ����װ�ã�������ȡ������ܽ��Ժ��ܶȵĴ�Сѡ���ռ�����ķ�����

��ϰ��ϵ�д�
�����Ŀ










