��Ŀ����
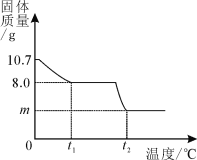
����Ŀ����6�֣��������岿��ʧˮ��õ�ij����ͭ���壨CuSO4��xH2O������ѩͬѧΪ�о�CuSO4��xH2O���Ⱥ��������ʵ���ɣ���������ʵ�飺ȡ10.7gCuSO4��xH2O�������ʹ��ֽ⣬���Ƴ�������������¶ȵı仯��ϵͼ�������ϣ�CuSO4��x H2O����Է�������Ϊ��160+18x��
��ͼ��ʾ����t1��ʱ�ù�����ȫʧȥ�ᾧˮ������ѧ����ʽΪ��CuSO4��xH2O ![]() CuSO4+ xH2O����
CuSO4+ xH2O����
��1����3�֣����������ͭ���壨CuSO4��xH2O����x��ֵ��
��2����2�֣�����ͭ������ͭԪ�ص����������Ƕ��٣���ȷ��0.1%��
��3����1�֣����µ�t2�������������Ǻ�ɫ���ʣ����ɫ������ ��
���𰸡���1��3
��2��29.9% ��2����
��3��CuO
��������
�����������1������ͼʾ��֪��0��t1ʱ�����ٹ�������Ϊ10.7 g -8.0 g =2.7g ����Ϊ����ˮ�����������û�ѧ����ʽ���м��㡣���岽�����£�
CuSO4��xH2O ![]() CuSO4+ xH2O��
CuSO4+ xH2O��
160 18x
8.0g 2.7g
160��8.0 g =18x��2.7g
x=3
��2������ͭ������ͭԪ�ص���������=64/(64+32+16��4+18) ��100%=29.9%
��3�����ݻ�ѧ��Ӧǰ��Ԫ������䣬��֪����ɫ����Ϊ����ͭ��

 ����ѵ�����⿼ϵ�д�
����ѵ�����⿼ϵ�д�����Ŀ����10�֣�������û����������ʱ������ˮ��������Ӧ���������£�����ˮ�����ܷ�Ӧ����һ�ֳ��������������һ�����塣С���ܺ��棬�������ʵ��̽��������ˮ������Ӧ��IJ��
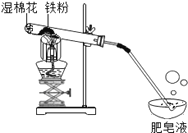
��1���Թ�β����һ��ʪ����Ŀ����_______________��
��2��̽�����ɵ�������ʲô��
��ȼ�ŵ�ľ�����������ݣ��б��������Ժ��з�����Ʈ�����С�˵�����ɵ�������_________��
��3��̽���Թ���ʣ�����ɷ���ʲô��
���������ϡ�
�������������� | FeO | Fe2O3 | Fe3O4 |
��ɫ��״̬ | ��ɫ��ĩ | ����ɫ��ĩ | ��ɫ���� |
�ܷ������� | �� | �� | �� |
��������֤���Թ���ʣ�����Ϊ��ɫ����ȫ��������������
����������衿����һ��ʣ�������Fe��Fe3O4���������ʣ�������_____________��
��ʵ��̽����
ʵ����� | ʵ�������� |
��ʵ����ۡ�����ˮ������Ӧ�Ļ�ѧ����ʽΪ___________________________________��
����˼�뽻�����ú�ɫ���岻������Fe2O3��������_______________________________��
�� ��չ �� �ѳ�����ұ������������Ҫ��Ӧԭ�����ڸ����£�һ����̼��ȡ����ʯ�������������ԭ��������д����ѧ��Ӧ����ʽ��______________________________________��