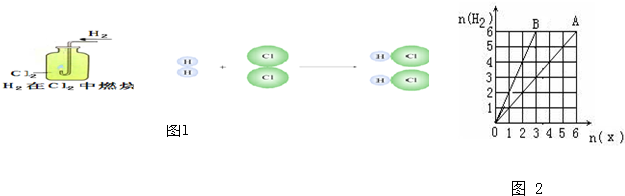
��Ŀ����
ij��ȤС���ͬѧ����ͼ��ʾװ����ȡ������Ȼ���һ��̽���й����������ʣ���ش�
��1��д�����Ϊ�ٺ͢ڵ����������ƣ��� �� �� ��
��2����д����ƿ�з�����Ӧ�Ļ�ѧ����ʽ�� ��
��3����ͼ��Bװ����ʢ����ˮ����ͭ��ɫ��ĩ����ɫ����ˮ����ͭ��ˮ������������йصĻ�ѧ����ʽΪ��
CuSO4���ף�+5H2O��CuSO4?5H2O��������Ԥ�Ȿʵ������У���ˮ����ͭ�Ƿ������� ����ǡ���

��1��д�����Ϊ�ٺ͢ڵ����������ƣ���
��2����д����ƿ�з�����Ӧ�Ļ�ѧ����ʽ��
��3����ͼ��Bװ����ʢ����ˮ����ͭ��ɫ��ĩ����ɫ����ˮ����ͭ��ˮ������������йصĻ�ѧ����ʽΪ��
CuSO4���ף�+5H2O��CuSO4?5H2O��������Ԥ�Ȿʵ������У���ˮ����ͭ�Ƿ�������

���㣺��������ȡװ��,�������ռ�����,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺���������ʵ�����Ʒ������顢�����뾻��
��������1��Ҫ��Ϥ�������������ơ���;��ʹ�÷�����
��2��ͨ������£����������ڶ������̵Ĵ������£��ֽ�����ˮ��������
��3���������������У������������ˮ������
��2��ͨ������£����������ڶ������̵Ĵ������£��ֽ�����ˮ��������
��3���������������У������������ˮ������
����⣺��1�����ǵ��ܣ������ڵ������������壻���Ǽ���ƿ���������ռ����壮
������ܣ�����ƿ��
��2�����������ڶ������̵Ĵ������·ֽ�Ļ�ѧ����ʽΪ��2H2O2
2H2O+O2����
���2H2O2
2H2O+O2����
��3����ʵ���У��������������У������������ˮ��������ʹ��ˮ����ͭ������
����ǣ�
������ܣ�����ƿ��
��2�����������ڶ������̵Ĵ������·ֽ�Ļ�ѧ����ʽΪ��2H2O2
| ||
���2H2O2
| ||
��3����ʵ���У��������������У������������ˮ��������ʹ��ˮ����ͭ������
����ǣ�
�����������ۺϿ�����ѧ���Ļ���ʵ���������漰��������;����ѧ����ʽ����д��ʵ�����������֪ʶ��ֻ���ۺ����⻯ѧ֪ʶ���ܹ���ȷ�Ľ��

��ϰ��ϵ�д�
�����Ŀ
һ����Һ������ֵ������ǣ�������
| A������ͬ�� | B����������治ͬ |
| C����ͬ�� | D�����Ƚ� |
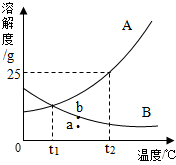 �������ͼ��A��B���ֹ������ʵ��ܽ�����ߣ��ش��������⣮
�������ͼ��A��B���ֹ������ʵ��ܽ�����ߣ��ش��������⣮