��Ŀ����
����������Һ��ϡ���ᷴӦ�Ļ�ѧ����ʽΪ____________��ʵ������һƿ��ǩ��ȱ��������Һ��Ϊ�˴��Բⶨÿ����������Һ������H2SO4�����ʵ�������λ��mol/mL������ʵ�鲽�����£�
�� ��С�ձ��м���10g��4%��NaOH��Һ��������������ˮϡ�ͣ��ٵ���2�� __________ ����ָʾ�����ƣ���Һ��
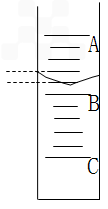
�� ��10mL��Ͳ��ע���������һ���̶ȡ���Ͳ��Һ���λ����ͼ��ʾ��A��B��B��C�̶ȼ�����1mL������̶�A����Ϊ8.0mL������Ͳ������������__________mL��
�� ��С�ձ��м���10g��4%��NaOH��Һ��������������ˮϡ�ͣ��ٵ���2�� __________ ����ָʾ�����ƣ���Һ��
�� ��10mL��Ͳ��ע���������һ���̶ȡ���Ͳ��Һ���λ����ͼ��ʾ��A��B��B��C�̶ȼ�����1mL������̶�A����Ϊ8.0mL������Ͳ������������__________mL��
���ý�ͷ�ι���ȡ��Ͳ�е����ᣬ��ε��������ձ��У�һֱ�ε���Һ��____________ɫǡ�ñ��__________ɫΪֹ����ʱ��Ͳ��Һ��Ķ���Ϊ2.2 mL
�� ����ȷ��ÿ����������Һ�к�����H2SO4�����ʵ�����д��������̣���
�� ����ȷ��ÿ����������Һ�к�����H2SO4�����ʵ�����д��������̣���
��2NaOH+H2SO4 ==Na2SO4+2H2O
�ڷ�̪
��7.2
�ܺ� ;��
�ݽ⣺�蹲��Ӧ������ x mol��
2NaOH + H2SO4 �� Na2SO4 + 2H2O
2mol 1mol
10��4%/40 x
2x= 10��4%/40
x=0.005mol
��Ӧ�й�����������Һ�����Ϊ��7.2-2.2=5.0mL
��ÿ������Һ�к�����H2SO4�����ʵ���Ϊ��0.005/5.0=0.001mol/mL
�ڷ�̪
��7.2
�ܺ� ;��
�ݽ⣺�蹲��Ӧ������ x mol��
2NaOH + H2SO4 �� Na2SO4 + 2H2O
2mol 1mol
10��4%/40 x
2x= 10��4%/40
x=0.005mol
��Ӧ�й�����������Һ�����Ϊ��7.2-2.2=5.0mL
��ÿ������Һ�к�����H2SO4�����ʵ���Ϊ��0.005/5.0=0.001mol/mL

��ϰ��ϵ�д�
 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
�����Ŀ