��Ŀ����
 ijʵ����������һ����������������KNO3��Һ����ʵ����ֻ�к�����NaCl��KNO3����ҩƷ��
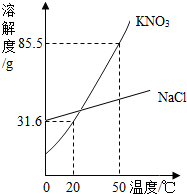
ijʵ����������һ����������������KNO3��Һ����ʵ����ֻ�к�����NaCl��KNO3����ҩƷ����1������KNO3��NaCl���ܽ��������ͼ.50��ʱ����85.5g����ҩƷ����100gˮ�У��õ�����KNO3��
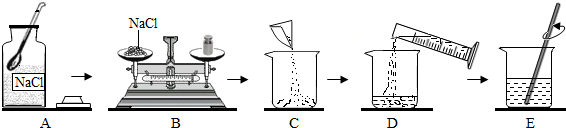
��2��С�����ᴿ���KNO3��������1000g������������Ϊ5%��KNO3��Һ������������ǣ����㡢
����⣬С��������Һ��KNO3������������ƫС����ԭ�������
��KNO3������Ȼ���� ���ܽ�ǰ���ձ���������ˮ
����ȡˮʱ�����Ӷ��� ��װƿʱ����������Һ������
��3��С�������Ҳ����һ����20��ʱ31.6%��KNO3��Һϡ������5%����Һ������Ϊ�Ƿ���У�
���㣺һ������������������Һ������,�����ܽ������������
ר�⣺��Һ����Һ���ܽ��
��������1�������ᴿ���ʵ��ܽ�����¶ȵĹ�ϵ���ǣ�
��2������������Һ�IJ����Լ���ɵ���������
��3������20��ʱKNO3���ܽ�ȣ������ʱ�����ʵ�����������һ�Ƚϣ�
��2������������Һ�IJ����Լ���ɵ���������
��3������20��ʱKNO3���ܽ�ȣ������ʱ�����ʵ�����������һ�Ƚϣ�
����⣺��1����KNO3��NaCl���ܽ������֪��50��ʱKNO3���ܽ��Ϊ85.5g�����Խ�85.5g����ҩƷ����100gˮ�У����ں������ʣ������γɵ��Dz�������Һ���ʴ�Ϊ�������ͣ�
��2��������Һ�IJ���Ϊ�����㡢�������ܽ⡢װƿ��������Һ��KNO3������������ƫС��ԭ��Ϊ�����ٻ��ܼ��࣬
KNO3������Ȼ�����������٣��ܽ�ǰ���ձ���������ˮ���ܼ��ࣻ����Ͳ��ȡˮʱ���Ӷ�������ȡ����Һ��ˮƫ�٣�����õ�����������ƫ��װƿʱ����������Һ����������Һ��������С��������������������ı䣬���Ԣ٢ڷ��ϣ��ʴ�Ϊ���������ܽ⣻�٢ڣ�
��3��20��ʱKNO3��Һ���ʵ���������Ϊ
%��24%�����Բ������31.6%��KNO3��Һ��������У�
��2��������Һ�IJ���Ϊ�����㡢�������ܽ⡢װƿ��������Һ��KNO3������������ƫС��ԭ��Ϊ�����ٻ��ܼ��࣬
KNO3������Ȼ�����������٣��ܽ�ǰ���ձ���������ˮ���ܼ��ࣻ����Ͳ��ȡˮʱ���Ӷ�������ȡ����Һ��ˮƫ�٣�����õ�����������ƫ��װƿʱ����������Һ����������Һ��������С��������������������ı䣬���Ԣ٢ڷ��ϣ��ʴ�Ϊ���������ܽ⣻�٢ڣ�
��3��20��ʱKNO3��Һ���ʵ���������Ϊ
| 31.6g |
| 31.6g+100g |
���������⿼�����ܽ�Ⱥ����������������йؼ��㣬�ѶȲ���������׳����ĵط���������������ƫС��ԭ��Ҫ������������������ʳ����ˣ��ܼ������ˣ�

��ϰ��ϵ�д�
 һŵ��ҵ�����ҵ���ּ�����������������ϵ�д�
һŵ��ҵ�����ҵ���ּ�����������������ϵ�д�
�����Ŀ
���л�ѧ����ʽ��д����ȷ���ǣ�������
A��CaCO3
| ||||
B��2KMnO4
| ||||
C��Al+O2
| ||||
| D��CO2+Ca��OH��2�TCaCO3��+H2O |
�����ƣ�Na2SO4���ǻ�ѧ�г�����һ�����ʣ�������Ԫ�صĻ��ϼ��ǣ�������
| A��+4 | B��6 | C��+6 | D��-2 |

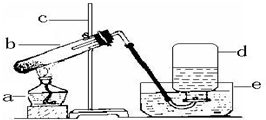 ��ͼΪʵ�����ü��ȸ�����صķ�����ȡ������װ��ͼ���ش����⣺
��ͼΪʵ�����ü��ȸ�����صķ�����ȡ������װ��ͼ���ش����⣺ �±���Ca��OH��2 ��NaOH���ܽ�����ݣ���ش��������⣺
�±���Ca��OH��2 ��NaOH���ܽ�����ݣ���ش��������⣺