��Ŀ����
�����벻����ѧ�������ʳ���dz����г��������ֻ�ѧ���ʡ�����һ����ɫ�������ʣ������ɴ��ʳ���е�һ�ֻ�����������ɡ�Ϊȷ������ɣ�ij��ѧ��ȤС���ͬѧ��ȡ�ð�ɫ����22.3g������̽��ʵ�飺
��һ�����ԣ�ʵ��Ŀ�ģ�ȷ����ɫ��������
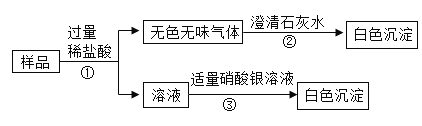
��ʵ�鷽������ͼ��ʾ
��ʵ����ۣ�
��1������ڷ�����Ӧ�Ļ�ѧ����ʽ��________��
��2������۷����ķ�Ӧ����________��Ӧ��ѡ����ϡ����ֽ⡱���û��������ֽ⡱ ֮һ����
��3���ù�����________��ѡ�һ���������ܡ�������֮һ������ NaCl������ж�������________��
��ʵ�鷴˼�����Է������ʵ����ʱ��ҩƷ�����ѡ���ҩƷ����������ʱ��Ӱ���������ɵ��жϣ����ԣ�ѡ��ҩƷʱһ��Ҫ���������Լ��Լ���Ӧ���ɵ����ʣ����������ʵļ���������š�
������������ʵ��Ŀ�ģ�ͨ�����ݷ�����ȷ����ɫ��������
��ʵ�����ݣ���ʵ��С���ͬѧ��ʵ�鲽�������������������Ͳ�����в����������������¼������������������ͼ��ʾ��
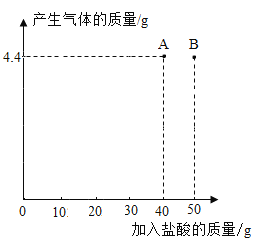
��4������������B��ʱ��������Һ�к��е�������______����д��ѧʽ����
��5����ͨ������ȷ���ð�ɫ���������ɷֵ�����_____��
 �żӾ���ϵ�д�
�żӾ���ϵ�д�