题目内容
(3分)向某铁粉样品中,加入溶质质量分数为10%的硫酸铜溶液160 g,恰好完全反应,样品中杂质不溶于水也不与硫酸铜反应。请结合下图提供的数据计算。
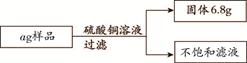
(1)a的数值。
(2)向不饱和滤液中加入40.8 g水,求所得溶液中溶质的质量分数.

(1)a的数值。
(2)向不饱和滤液中加入40.8 g水,求所得溶液中溶质的质量分数.
【解】设:样品中铁的质量为x,生成铜的质量为y,生成硫酸亚铁的质量为z[
Fe + CuSO4 = FeSO4 + Cu
56 160 152 64
x 160g×10% z y
x = 5.6g y = 6.4g z = 15.2g ···························1 分
(1)a =" 5.6g" +(6.8g – 6.4g)= 6g ····························1 分
(2)5.6g + 160g – 6.4g + 40.8g = 200g
15.2g / 200g × 100% = 7.6% ···························1 分
答:(1)a的数值为6.
(2)所得溶液中溶质的质量分数为7.6%
Fe + CuSO4 = FeSO4 + Cu
56 160 152 64
x 160g×10% z y
x = 5.6g y = 6.4g z = 15.2g ···························1 分
(1)a =" 5.6g" +(6.8g – 6.4g)= 6g ····························1 分
(2)5.6g + 160g – 6.4g + 40.8g = 200g
15.2g / 200g × 100% = 7.6% ···························1 分
答:(1)a的数值为6.
(2)所得溶液中溶质的质量分数为7.6%
试题分析:发生的反应为Fe + CuSO4 = FeSO4 + Cu,已知硫酸铜的质量为160g×10%=16g;结合化学方程式求出样品中铁的质量,生成铜的质量和生成硫酸亚铁的质量;再结合题意及溶质质量分数的计算公式,即可求得.

练习册系列答案
 名校课堂系列答案
名校课堂系列答案
相关题目