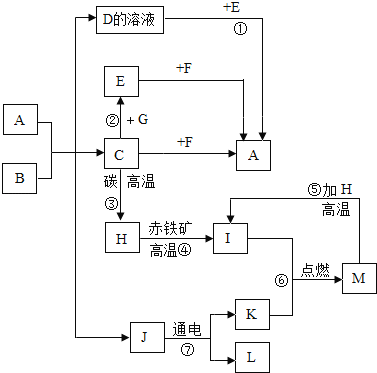
��Ŀ����
����Ŀ����6�֣��±���Ca(OH)2��NaOH���ܽ����������ش�����������
�¶�/�� | 0 | 20 | 40 | 60 | 80 | 100 | |
�ܽ��/g | Ca(OH)2 | 0.19 | 0.17 | 0.14[ | 0.12[ | 0.09 | 0.08 |
NaOH | 31 | 91 | 111 | 129 | 313 | 336 | |
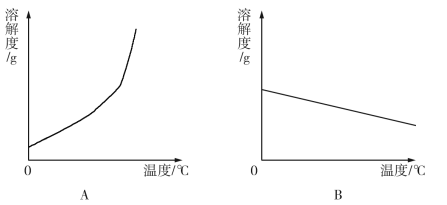
��1�������ϱ����ݣ�����Ca(OH)2��NaOH���ܽ�����ߣ���ͼ���ܱ�ʾNaOH�ܽ�����ߵ��� ����A��B����
��2��Ҫ���һƿ�ӽ����͵�Ca(OH)2��Һ��ɱ�����Һ�������ʩ�У�
�����������������������¶����������¶���������ˮ��������ˮ�����ָ���ԭ�¶�����������ʯ����
���д�ʩ��ȷ���� ��
A���ڢܢ� B���ۢ� C���٢ۢݢ� D���٢ڢݢ�
��3��20��ʱ��191g ����NaOH��Һ������10gˮ���ٽ��µ�20����������NaOH���������Ϊ ��
��4������20��ʱCa(OH)2�ı�����Һ������Һ���������м���һ����CaO��õ�����Һ������Һ������ʱ��Һ������������������ �ף�����������������������������
��5������60��ʱ��Ca(OH)2��NaOH�������ʵı�����Һ����Ҫ�õ��ϴ�����NaOH���壬Ӧ��ȡ������������ ��
��6��20��ʱ�����ⶨNaOH��Һ��pH�����Ƚ�pH��ֽ������ˮ��ʪ���������ⶨ����������Һ��pH ������ƫ��������ƫС����������Ӱ��������
��������1��A �� ��2��D �� ��3��9.1 g ��©д��λ���÷��� �� ��4���� ��
�й���5�����½ᾧ������ �� ��6��ƫС��
��������
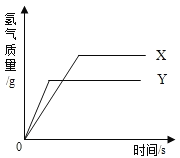
�����������1���������Ƶ��ܽ�����¶ȵ����߶����ӣ������ܱ�ʾ���������ܽ�����ߵ���ͼA��
��2���������Ƶ��ܽ�����¶ȵ����߶����ͣ�����Ҫʹ�������ƵIJ�������Һ��Ϊ������Һ���ɲ�ȡ�Ĵ�ʩΪ�����������������ƻ�����ˮ�ֻ������¶ȣ�����̼����ʱ�������ƺ�ˮ��Ӧ�����������ƣ��ܼ����������٣������¶����ߣ�����Ҳ����ʹ�����͵�����������Һ��Ϊ������Һ���ʴ�ΪD��
��3��20��ʱ���������Ƶ��ܽ����91g ,�� 100g����ܽ�91g�������ƣ�������10gˮ�������������Ƶ�����Ϊ9.1g��
��4���������Ƶ��ܽ�����¶ȵ����߶����ͣ�����������ˮ�����������Ƶķ�Ӧ�ų��������ȣ�������20��ʱCa(OH)2�ı�����Һ������Һ���м���һ����CaO��õ�����Һ������Һ������ʱ��Һ�����������������ǣ���<����
��5���������Ƶ��ܽ�����¶ȵ����߶����ͣ��������Ƶ��ܽ�����¶ȵ����߶��������������Ƶ��ܽ�Ⱥ�С�����Կ��ý��£���ȴ�ȱ�����Һ�ķ���ϡ�������ƽᾧ������Ȼ����˼��ɵõ���Ϊ�������������ƣ�
��6��20��ʱ�����ⶨNaOH��Һ��pH�����Ƚ�pH��ֽ������ˮ��ʪ���������ⶨ����������Һ����������������С�����Լ�����pHֵƫС��

 �����������һ��һ��ϵ�д�
�����������һ��һ��ϵ�д�����Ŀ����������Ҫ�Ľ������ϣ��ڹ�ũҵ������������Ӧ�÷dz��㷺��
��������Ӧ��
��1����������Ʒ������������������йص���________________������ĸ����ͬ����
A����ǯ B������ C������
��2�������צ�-Fe������������ʳƷ���ʣ���֮Ϊ��˫����������Ϊ�������տ����е�________________��
��������ұ��
��ҵ������ԭ���Ǹ�������CO����ԭ�����������������л�ԭ��������ش��������⣺
��1��д���Գ�����Ϊԭ�ϣ��ڸ����������Ļ�ѧ����ʽ��___________________________��
��2����¯�����У���̿�����ó��˿�������һ����̼�⣬����_____________________��
����ʵ��̽������ԭ��
ij��ѧ��ȤС����ʵ������ģ�ҵ������ԭ������̽��CO��Fe2O3��Ӧ��IJ��
ͨ����������֪���������ᣨH2C2O4��������Ũ�����ϼ��Ȼ����һ����̼����Ӧ����ʽΪ��H2C2O4 ![]() CO��+ CO2��+ H2O��
CO��+ CO2��+ H2O��
��NaOH��Һ�������ն�����̼����Ӧ����ʽΪ��2NaOH+CO2=Na2CO3+H2O
�������£�Ca(OH)2����ˮ���������������ͼ��ʵ��װ�ã����װ�ûش��������⣺
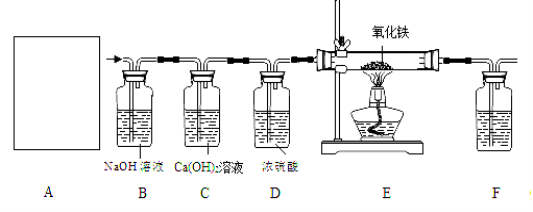
��1��ͼA��������������Ũ������ȡCO�ķ�Ӧװ�ã�����ΪӦѡ����ͼ�е�_________����װ�ñ�ţ���
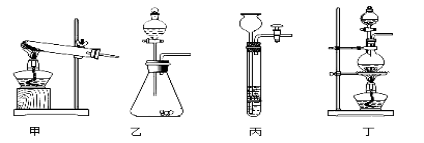
��2��ͼ��װ��C��D�����÷ֱ��� ____________��_______________��
��3��Ϊ��֤���������ж�����̼��װ��F�����Լ�Ӧ����������_____________�����Լ����ƣ�����Ӧ�Ļ�ѧ����ʽΪ ______________________��
��4����װ�������һ������ȱ�ݣ�����Ϊ��______________________________��
��5��ʵ��ʱȷ��ȡһ������������Fe2O3���尴��ͼ����ʵ�飬��E�й���ȫ����ں���ͨ��COֱ����������ȴ�����õ���ɫ�Ĺ�����뵽������ϡ���ᣬ���ֹ���ȫ���ܽ⣬�������ݲ�����
�������ϣ�a. ������������������ϡ�����о���ȫ���ܽ⡣
b. Fe2O3��CO��Ӧ�Ĺ������������������£�
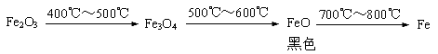
���ݡ������ݡ����룬��ɫ��ĩ�����ǣ��� Fe3O4 ����_________ �� ____________��
��6���������� �õ�����ƽ�����������������£�
���������� | ��������������������� | װ��F���������ʵ������� | |
��Ӧǰ | 28.20 g | 33.00 g | 300.0 g |
��Ӧ�� | 32.84 g | 300.4 g |
�����������ݣ�������Fe2O3��������Ϊ_____________g����ѡ����Ч�����ݣ��ƶϳ���Ӧ���ɫ����ijɷ֣�д��������̣�__________________________��
�����ƶϽ����д����ʵ���в������������Ļ�ѧ��Ӧ����ʽ��__________________________��