��Ŀ����
����Ŀ���������������������й㷺��Ӧ�á�
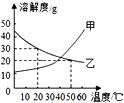
(1)���н�����Ʒ�У����ý��������Ե���______(����ĸ���)��

(2)���������������______��
(3)��ҵ����һ����̼�ͳ�����������Ӧ�Ļ�ѧ����ʽΪ__________________________��
(4)���÷Ͼɵ��ͭñ(�� Cu��Zn)��ȡ����ͭ(Cu)�����õ�����п��Һ����Ҫ��������(��Ӧ��������ȥ)��

��֪��2Cu+2H2SO4+2O2=CuSO4+2H2O
�ٹ��̢��з�������������� ________��
�ڹ��̢����������������Ӧ�Ļ�ѧ����ʽΪ_____��
��A~E �к�ͭ��п����Ԫ�ص������� ________(����ĸ���)��
���𰸡� C ��ˮ������ͬʱ�Ӵ� 3CO + Fe2O3 ���� 2Fe+3CO2 ���� Zn+H2SO4=ZnSO4+H2�� A C
����������1��A���ƽ���Ʒ�����˻ƽ�Ľ��������뵼�����أ�����
B�����������������ĵ����ԣ�����
C��ͭ����������ͭ�ĵ����ԣ���ȷ����ѡC��
��2������������е�ˮ�������Ӵ��Ϳ���������
��3��һ����̼���������ڸ��µ������·�Ӧ�������Ͷ�����̼���ʷ���ʽΪ3CO + Fe2O3 ���� 2Fe+3CO2
��4���ٲ�����ǽ���Һ����IJ��裬��Һ�ǹ��ˣ�
�ڲ������п��ϡ���ᷴӦ��������п����������Ӧ����ʽΪZn+H2SO4=ZnSO4+H2��
����ҺA�е�п��ϡ���ᷴӦ��������п���ɣ�ͭ��ϡ�����������Ӧ��������ͭ������ҺA�к�������п������ͭ����A��ȷ������B�ǹ�����п���ʹ���C�к���ͭ��п����ȷ������D��ֻ������ͭ������E��ֻ������п����ѡAC��