��Ŀ����
ij��ͬѧ�����ꡰ������̼����ȡ�����ʡ�ʵ���Һ����ʢ�д������������Ȼ��ƵĻ����Һ���������������ʣ���Ϊ�˶Է�Һ����̽����ijС��ͬѧ����������ʵ�飺ȡ��Һ���еij�����Һ20gʢ���ձ��У���μ���5.3%��̼������Һʹ����ȫ��Ӧ���й�ʵ�����ݼ�¼���±�����̼������Һ�μ���50g���ѹ�����ʱ�����������ʽ��й��˺�ø������1.0g���������ݼ�¼���£�| �ⶨ���� | ��1�� | ��2�� | ��3�� | ��4�� | ��5�� |
| ����Na2CO3��Һ/g | 10.0 | 20.0 | 30.0 | 40.0 | 50.0 |
| �������ʵ�������/g | 29.78 | 39.56 | 49.56 | 59.56 | δ�� |
��2��20g��Һ�к��Ȼ����������
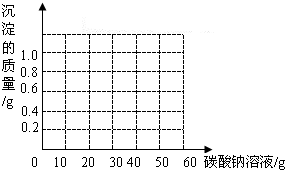
��3��������ͼ�л������ɳ��������Na2CO3��Һ�����仯�����ߣ�
��4������Һ���������ʸպ��������ʱ��������Һ���Ȼ��Ƶ�����������
��������1�����ݱ���ʵ�����ݣ���������̼������Һ�ļ����ձ����������������ı仯��������̼������Һ���ձ���ʣ�������������ټ��٣�����Һ��������ȫ��Ӧ�����ô�ʱ�仯ǰ��ʣ�����������IJ��������ɶ�����̼��������
��2����̼���������ᷴӦ�Ļ�ѧ����ʽ���ɷų�������̼���������㷴Ӧ�����������������
��3���ɱ���ʵ�����ݣ��ж�������̼������Һ���Ȼ��ƿ�ʼ��Ӧ���ɳ���ʱ����������Ϊ���ߵ���㣻�ɷ�Ӧ��ѧ����ʽ�������ɳ���������������ǡ����ȫ��Ӧʱ������̼������Һ���������ж����ߵ��۵㣻
��4����Ӧ����ҺΪ�Ȼ�����Һ�������Ȼ�������Ϊ����̼���������ἰ�Ȼ��Ʒ�Ӧ�������Ȼ��Ƶ������ͣ���Ӧ��������Һ�������ɸ��������غ㶨����ã����������Һ������������������������Һ���Ȼ��Ƶ�����������
��2����̼���������ᷴӦ�Ļ�ѧ����ʽ���ɷų�������̼���������㷴Ӧ�����������������
��3���ɱ���ʵ�����ݣ��ж�������̼������Һ���Ȼ��ƿ�ʼ��Ӧ���ɳ���ʱ����������Ϊ���ߵ���㣻�ɷ�Ӧ��ѧ����ʽ�������ɳ���������������ǡ����ȫ��Ӧʱ������̼������Һ���������ж����ߵ��۵㣻
��4����Ӧ����ҺΪ�Ȼ�����Һ�������Ȼ�������Ϊ����̼���������ἰ�Ȼ��Ʒ�Ӧ�������Ȼ��Ƶ������ͣ���Ӧ��������Һ�������ɸ��������غ㶨����ã����������Һ������������������������Һ���Ȼ��Ƶ�����������
����⣺��1����ʵ�����ݣ���1�Ρ���2�μ���̼������Һʱ���ձ���ʣ��������������������С������3�μ���̼������Һ���ձ�����ʣ�������������ټ��٣�˵��ǰ�����������̼������Һ�Ѿ���������ȫ��Ӧ����ʱ��Ӧ�ų�������̼������=20g+20g-39.56g=0.44g��
��2����20g��Һ�к��Ȼ��������Ϊx
Na2CO3+2HCl�T2NaCl+H2O+CO2��
73 44
x 0.44g
=
x=0.73g
��3��������20g̼������Һʱ�������Һ�е�������ȫ��Ӧ����ʱ���������̼������Һ��ʼ���Ȼ��Ʒ�Ӧ������̼��Ƴ�������ʱΪ���ߵ���㣻
��ǡ����ȫ��Ӧʱ������̼������Һ������Ϊy
Na2CO3+CaCl2�T2NaCl+CaCO3��
106 100
y��5.3% 1g
=
y=20g��������̼������Һ��������20+20��g=40gʱ��ǡ����ȫ��Ӧ����ʱΪ���ߵ��۵㣬�Ժ�����������ٸı䣻
��4�������40g̼������Һʱ���������Ȼ��Ƶ�����Ϊz
Na2CO3��2NaCl
106 117
40g��5.3% z
=
z=2.34g
������Һ���Ȼ��Ƶ���������=
��100%=4.0%
�ʴ�Ϊ��
��1��������ȫ��Ӧʱ���ɵĶ�����̼������Ϊ0.44g��
��2��20g��Һ�к��Ȼ��������Ϊ0.73g��
��3��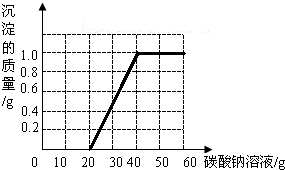
��4��������Һ���Ȼ��Ƶ���������Ϊ4.0%��
�������ⷨ������ȷ�����֣�
��2����20g��Һ�к��Ȼ��������Ϊx
Na2CO3+2HCl�T2NaCl+H2O+CO2��
73 44
x 0.44g
| 73 |
| x |
| 44 |
| 0.44g |
��3��������20g̼������Һʱ�������Һ�е�������ȫ��Ӧ����ʱ���������̼������Һ��ʼ���Ȼ��Ʒ�Ӧ������̼��Ƴ�������ʱΪ���ߵ���㣻
��ǡ����ȫ��Ӧʱ������̼������Һ������Ϊy
Na2CO3+CaCl2�T2NaCl+CaCO3��
106 100
y��5.3% 1g
| 106 |
| y��5.3% |
| 100 |
| 1g |
��4�������40g̼������Һʱ���������Ȼ��Ƶ�����Ϊz
Na2CO3��2NaCl
106 117
40g��5.3% z
| 106 |
| 40g��5.3% |
| 117 |
| z |
������Һ���Ȼ��Ƶ���������=
| 2.34g |
| 59.56g-1g |
�ʴ�Ϊ��
��1��������ȫ��Ӧʱ���ɵĶ�����̼������Ϊ0.44g��
��2��20g��Һ�к��Ȼ��������Ϊ0.73g��
��3��

��4��������Һ���Ȼ��Ƶ���������Ϊ4.0%��
�������ⷨ������ȷ�����֣�
���������������̼������Һ�������Ӷ����ɳ�������������ʱ�����ߵ���㼴�����Һ������ǡ����ȫ��Ӧ�����ߵ��۵㼴�����Һ���Ȼ���ǡ����ȫ��Ӧ��

��ϰ��ϵ�д�
�����Ŀ
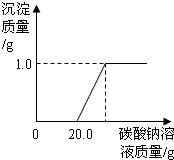 ij��ͬѧ�����ꡰ������̼����ȡ�����ʡ�ʵ���Һ����ʢ�д������������Ȼ��ƵĻ����Һ���������������ʣ���Ϊ�˶Է�Һ���д�����ijͬѧ��������ʵ�飺ȡ��Һ���ϲ���Һ20.0g���ձ��У���ε���������������Ϊ5.3%��̼������Һ������������̼������Һ������/g�������ɳ�����������/g���ı仯��ϵ��ͼ��ʾ������������ȷ��0.1%��
ij��ͬѧ�����ꡰ������̼����ȡ�����ʡ�ʵ���Һ����ʢ�д������������Ȼ��ƵĻ����Һ���������������ʣ���Ϊ�˶Է�Һ���д�����ijͬѧ��������ʵ�飺ȡ��Һ���ϲ���Һ20.0g���ձ��У���ε���������������Ϊ5.3%��̼������Һ������������̼������Һ������/g�������ɳ�����������/g���ı仯��ϵ��ͼ��ʾ������������ȷ��0.1%�� ��2006?������һģ��ij��ͬѧ�����ꡰ������̼����ȡ�����ʡ�ʵ���Һ����ʢ�д������������Ȼ��ƵĻ����Һ���������������ʣ���Ϊ�˶Է�Һ���д�����ijͬѧ��������ʵ�飺ȡ��Һ�����ϲ���Һ40mL���ձ��У���ε���Na2CO3��Һ����������¼����Na2CO3��Һ������x�����й����ı仯��ϵ��ͼ��ʾ��ͼ�������꣨y����ʾ
��2006?������һģ��ij��ͬѧ�����ꡰ������̼����ȡ�����ʡ�ʵ���Һ����ʢ�д������������Ȼ��ƵĻ����Һ���������������ʣ���Ϊ�˶Է�Һ���д�����ijͬѧ��������ʵ�飺ȡ��Һ�����ϲ���Һ40mL���ձ��У���ε���Na2CO3��Һ����������¼����Na2CO3��Һ������x�����й����ı仯��ϵ��ͼ��ʾ��ͼ�������꣨y����ʾ ij��ͬѧ�����ꡰ������̼����ȡ�����ʡ�ʵ���Һ����ʢ�д������������Ȼ��ƵĻ����Һ���������������ʣ���Ϊ�˶Է�Һ���д�����ijͬѧ��������ʵ�飺ȡ��Һ���ϲ���Һ20.0g���ձ��У���ε���������������Ϊ5.3%��̼������Һֱ������������̼������Һ�����������ɳ����������ı仯��ϵ��ͼ��ʾ��
ij��ͬѧ�����ꡰ������̼����ȡ�����ʡ�ʵ���Һ����ʢ�д������������Ȼ��ƵĻ����Һ���������������ʣ���Ϊ�˶Է�Һ���д�����ijͬѧ��������ʵ�飺ȡ��Һ���ϲ���Һ20.0g���ձ��У���ε���������������Ϊ5.3%��̼������Һֱ������������̼������Һ�����������ɳ����������ı仯��ϵ��ͼ��ʾ��