��Ŀ����
����Ŀ�����������벻����Դ����Դ��Ӫ�����ʡ�������ѧ֪ʶ�ش���������
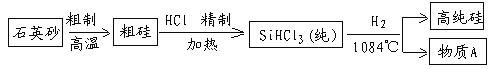
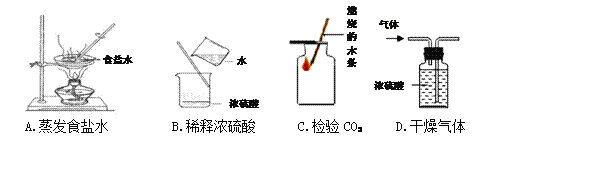
(1)��ͼ��ʾ̩���м���ȼ��ʹ�õķ�չ����(������Ϊ��Ҫ�ɷ�)
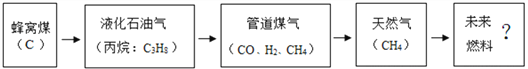
�����������������������˵��������Ϊδ��������Դ���ŵ�֮һ____________________��
�ڱ���ȼ����ʾ��ͼ����ͼ��ʾ
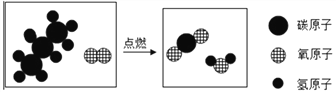
�÷�Ӧǰ�����������ԭ�ӡ�_________��________(�ѧ����)���÷�Ӧ�Ļ�ѧ����ʽΪ____________________________��
(2)ˮ������֮Դ�������������Ҫ���塣
�ٱ��ˮ����Ԫ�صĻ��ϼ�__________��
�ڷ�������ʱ������Ա���ø�ѹˮǹ������Ҫԭ����_________________��
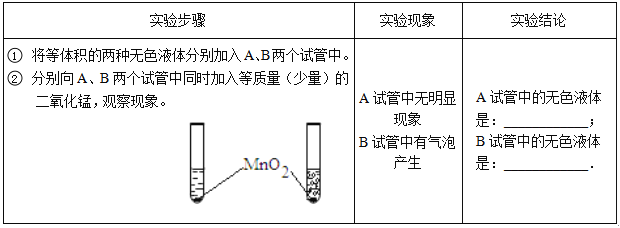
����Ȼˮ���ΪӲˮ������Ϊ���к��н϶�ĸ����Ӻ�þ���ӣ���þ�������û�ѧ���ű�ʾΪ__________���ճ������п���________________�ķ�������ˮ��Ӳ�ȡ�
(3)����ʳΪ�죬�й�����ʳ�Ļ������
������ʳ���и��������ʵ���__________(��д��ĸ���)��
A���� B������ C��ƻ�� D�����
���������д��������١�������������0.01%��Ԫ�س�Ϊ����Ԫ����������ÿ�ն���Ԫ�ص���Ҫ����С��ȴ�ز����١������������������Ԫ�ص���_________��
A���� B���� C��Ǧ D����
���𰸡� ԭ����Դ�㡢��������Ⱦ��ȼ����ֵ�� C H C3H8 + 5O2 ��ȼ 3CO2 + 4H2O ![]() ���¶Ƚ��͵��Ż������ Mg2+ ��� B AD
���¶Ƚ��͵��Ż������ Mg2+ ��� B AD
��������(1)�ٸ���ԭ����Դ�㡢��������Ⱦ��ȼ����ֵ�߽�𣻢���ʾ��ͼ��������غ㶨�ɽ��(2)�ٸ��ݻ��ϼ۵ı�ʾ��������Ԫ�ط������Ϸ�������ϼۡ�������ǰ�������ں�������Ϊ1ʱ������ʡ�Խ�𣻢ڸ������ԭ��������𣻢۸��ݸ������ӵı�ʾ�������ڱ�ʾ�����ӵ�Ԫ�ط��Ż�ԭ���ŵ����Ͻǣ���������������������������������ǰ�����������ں�1�����ʱ��1Ҫʡ�Խ�𣻸�������������еķ�������ˮ��Ӳ�Ƚ��(3)��ʳ������������ҪӪ���������жϣ��ڸ��������������Ԫ���У������ܡ�ͭ��п�������̡��⡢�����⡢�����(1)��������Ϊδ��������Դ���ŵ�֮һ�Dz�������Ⱦ����ԭ����Դ���ȼ����ֵ�ߣ����ڸ��������غ㶨�ɿ�֪����ѧ��Ӧǰ��ԭ�ӵ��������Ŀ���䡣��Ϸ�Ӧ����ʾ��ͼ��֪���÷�Ӧǰ�����������ԭ�ӡ�C��H(�ѧ����)���÷�Ӧ�Ļ�ѧ����ʽΪC3H8+5O2![]() 3CO2+4H2O��(2)�ٸ��ݻ��ϼ۵ı�ʾ��������Ԫ�ط������Ϸ�������ϼۡ�������ǰ�������ں�������Ϊ1ʱ������ʡ�ԡ�ˮ����Ԫ����-2�ۣ�����Ԫ����+1�ۣ�ˮ����Ԫ�صĻ��ϼۿɱ�ʾΪ
3CO2+4H2O��(2)�ٸ��ݻ��ϼ۵ı�ʾ��������Ԫ�ط������Ϸ�������ϼۡ�������ǰ�������ں�������Ϊ1ʱ������ʡ�ԡ�ˮ����Ԫ����-2�ۣ�����Ԫ����+1�ۣ�ˮ����Ԫ�صĻ��ϼۿɱ�ʾΪ![]() ����ˮ�ܽ����¶ȡ��ʷ�������ʱ������Ա���ø�ѹˮǹ������Ҫԭ���ǽ��¶Ƚ��͵���ȼ���Ż�����£������ӵı�ʾ�������ڱ�ʾ�����ӵ�Ԫ�ط��Ż�ԭ���ŵ����Ͻǣ���������������������������������ǰ�����������ں�1�����ʱ��1Ҫʡ�ԡ���þ���ӿɱ�ʾΪMg2+��(3)��A��������������ҪӪ���������࣬����B����������������ҪӪ�����ǵ����ʣ���ȷ��C��ƻ������������ҪӪ������ά���أ�����D���������������ҪӪ������ά���أ�����ѡB���������������Ԫ���У������ܡ�ͭ��п�������̡��⡢�����⡢������ѡAD��
����ˮ�ܽ����¶ȡ��ʷ�������ʱ������Ա���ø�ѹˮǹ������Ҫԭ���ǽ��¶Ƚ��͵���ȼ���Ż�����£������ӵı�ʾ�������ڱ�ʾ�����ӵ�Ԫ�ط��Ż�ԭ���ŵ����Ͻǣ���������������������������������ǰ�����������ں�1�����ʱ��1Ҫʡ�ԡ���þ���ӿɱ�ʾΪMg2+��(3)��A��������������ҪӪ���������࣬����B����������������ҪӪ�����ǵ����ʣ���ȷ��C��ƻ������������ҪӪ������ά���أ�����D���������������ҪӪ������ά���أ�����ѡB���������������Ԫ���У������ܡ�ͭ��п�������̡��⡢�����⡢������ѡAD��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
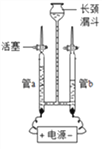
Сѧ��10����Ӧ����ϵ�д�����Ŀ��Ǧ������������������ʹ�ù㷺���乹��ʾ��ͼ��ͼ1���ش��������⣺
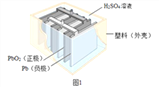
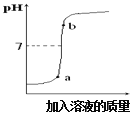
(1)ͼ1Ǧ���طŵ�����У���Ӧ�Ļ�ѧ����ʽΪ Pb+PbO2+2H2SO4�T2PbSO4+2H2O���ݴ˿�֪��Ǧ�����ڷŵ�ʱ����Һ��pH����_____(����������������С������������)��
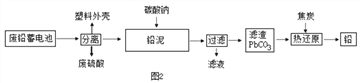
(2)��ͼ2�ǻ��շ�Ǧ���ص�һ�ֹ������̡�
��д����̿�Ȼ�ԭ����Ǧ�Ļ�ѧ����ʽ�� ________________________
�ڽ�����ͼ�еķ��������Һ��һ��������ϣ��ٽ����õĻ��Һ��____�����½ᾧ�����˵Ȳ�������������Na2SO4��10H2O���塣
�������һЩ�����۵�����ݣ�
���� | �� | Ǧ | �� | �� |
�۵��M�� | 231.9 | 327.5 | 271.3 | 320.9 |
�ճ����ñ���˿���顢Ǧ�������ӵȽ�����ɣ����۵�ԼΪ________________��
A�� 231.9-327.5�� B��271.3-320.9�� C��60-80�� D��20-40��
�ܷ�����ֱ���ŷŻ���Ⱦ��������ѡ����������кͺ����ŷţ�
���� | CaCO3 | NH3 | Ca(OH)2 |
�г��ο���(Ԫ/kg ) | 1.8 | 6.5 | 2.0 |
��֪��2NH3+H2SO4�T(NH4)2SO4�����Ҫ�����ٵ�Ǯ���к͵���������Ũ�ȵķ����ᣬ��Ӧѡ��_________ ��