��Ŀ����
ij��ѧ��ȤС��ʹ����ͼ��ʾװ�ã���ij����ͨ�Ͻ�ijɷֽ��в�������ȡ����ϡ�������ձ��У��������м���14.0g�Ͻ���Ʒ��ʼ��ʱʱ������������ƽ�Ķ�����¼���±��У���ش��������⣺
��1���÷�Ӧ��������������Ϊ
��2���Ͻ���ͭ����������Ϊ���٣�
��3�����ݵ�����ƽ������ʱ��Ĺ�ϵ�������÷�Ӧ���й����У������IJ���������ʲô�ص㣿
��
| ���ձ� | ���� ����� | �������ۺ� 5���� | �������ۺ� 12���� | �������ۺ� 21���� | �������ۺ� 40���� | �������ۺ� 3Сʱ | |
| ������g�� | 28.5 | 169.7 | 183.6 | 183.5 | 183.4 | 183.3 | 183.3 |
��2���Ͻ���ͭ����������Ϊ���٣�
��3�����ݵ�����ƽ������ʱ��Ĺ�ϵ�������÷�Ӧ���й����У������IJ���������ʲô�ص㣿
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
��������ͭ�Ͻ���������ϡ���ᷴӦ�ų����������������غ㶨�ɣ����ٵ��������������������������������������������Ӧ���������������������ͭ�Ͻ���ͭ�����������ɽ��
����⣺��1����������������Ϊ��169.7g+14.0g-183.3g=0.4g���𰸣�0.4g��
��2����Ͻ���Fe������Ϊx�����ɵ�FeSO4����Ϊy��
Fe+H2SO4=FeSO4+H2��
56 2
x 0.4g
=
��
x=11.2g��
�Ͻ���ͭ����������Ϊ��
��100%=20%��
�𣺺Ͻ���ͭ����������Ϊ80%��
��3����Ϊ�����Ũ��Խ��ӦԽ�죬���ŷ�Ӧ�Ľ��У������Ũ�ȼ�С����Ӧ�ٶȱ��������������������ٶ��ɿ������
�𰸣������Ũ��Խ��ӦԽ�죬���ŷ�Ӧ�Ľ��У������Ũ�ȼ�С����Ӧ�ٶȱ��������������������ٶ��ɿ������
��2����Ͻ���Fe������Ϊx�����ɵ�FeSO4����Ϊy��
Fe+H2SO4=FeSO4+H2��
56 2
x 0.4g
| 56 |
| x |
| 2 |
| 0.4g |
x=11.2g��
�Ͻ���ͭ����������Ϊ��
| 14.0g-11.2g |
| 14g |
�𣺺Ͻ���ͭ����������Ϊ80%��
��3����Ϊ�����Ũ��Խ��ӦԽ�죬���ŷ�Ӧ�Ľ��У������Ũ�ȼ�С����Ӧ�ٶȱ��������������������ٶ��ɿ������
�𰸣������Ũ��Խ��ӦԽ�죬���ŷ�Ӧ�Ľ��У������Ũ�ȼ�С����Ӧ�ٶȱ��������������������ٶ��ɿ������
���������ݱ����ṩ����Ϣ�����жϷ�Ӧ���е��������������ƽ�������ٱ仯ʱ��˵���Ѿ���ȫ��Ӧ������������������������������������һ�����������������

��ϰ��ϵ�д�
 ��˼ά������ҵ��ټ��ִ�ѧ������ϵ�д�
��˼ά������ҵ��ټ��ִ�ѧ������ϵ�д� �����������Ż�ѧϰϵ�д�
�����������Ż�ѧϰϵ�д�
�����Ŀ
����CO2��CO���ɲ��õķ����ǣ�������
�ٷֱ�ͨ����ɫʯ����Һ�� �ڷֱ���ȼ�ŵ�ľ�� �۷ֱ�ͨ��ʯ��ˮ�� �ֱܷ�ͨ�����ȵ�����ͭ��
�ٷֱ�ͨ����ɫʯ����Һ�� �ڷֱ���ȼ�ŵ�ľ�� �۷ֱ�ͨ��ʯ��ˮ�� �ֱܷ�ͨ�����ȵ�����ͭ��
| A���ڢۢ� | B���ڢ� |
| C���٢ڢۢ� | D���ۢ� |

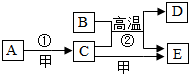 ��ͼA��EΪ����֮���ת����ϵ������ת������δ��������֪��AΪ��ɫ���嵥�ʣ�BΪ��ɫ��ĩ��A��D�γɵĺϽ���������������ʹ�����Ľ������ϣ���Ϊ���������嵥�ʣ��밴Ҫ��������пհף�
��ͼA��EΪ����֮���ת����ϵ������ת������δ��������֪��AΪ��ɫ���嵥�ʣ�BΪ��ɫ��ĩ��A��D�γɵĺϽ���������������ʹ�����Ľ������ϣ���Ϊ���������嵥�ʣ��밴Ҫ��������пհף�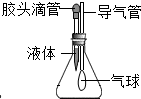 ��ͼ��ʾ��ͷ�ι���������������Һ����ƿ���Ƕ�����̼���壮
��ͼ��ʾ��ͷ�ι���������������Һ����ƿ���Ƕ�����̼���壮