��Ŀ����
����Ŀ������������һ����Ҫ�Ļ���ԭ�ϣ�Ҳ��ʵ�����г�����һ���Լ����������Ʊ����ܷⱣ�棬��Ϊ��������ж�����̼������ѧ��Ӧ�����ʣ��û�ѧ����ʽ��ʾ����ԭ��__________��
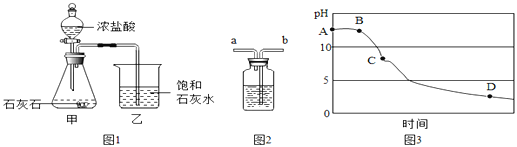
�����Ǽ�λͬѧ��һƿ�������ƹ���ɷֽ��е�̽����
̽��һ��ȷ��һƿ�������ƹ����Ƿ����
Сǿ������
ʵ����� | ʵ������ | ���� |
ȡ������Ʒ���Թ��У�����һ����ˮ����Ʒȫ�ܽ�μ�������__________����һ����Һ���ƣ� | �����ݲ��� | ___________ |
̽������ȷ���������ƹ�����ʵij̶�
С�շ�����
ʵ����� | ʵ������ | ���� |
��ȡ������Ʒ���Թ��У�����һ����ˮ����Ʒȫ�ܽ�μӹ������Ȼ�����Һ | ������ɫ���� | �������ƹ��岿�� ���� |
�ھ��ú�ȡ�ϲ���Һ���Թ��У�����2�η�̪��Һ | _________ |
������ʵ���������ͬѧ�����������������Һ�����Ȼ�����Һ������Ϊ�÷���_____ (����С������С�)�����ɣ�________̽�����������������ֱ��ʵ��������ƹ������������Ƶ���������
��1����������С����С����λͬѧ��ͬ�����ʵ�鷽����
ͨ��С��ʵ�鷽���������Ʒ���������Ƶ���������________��д��������̣�
��2����С��ʵ�鲽������
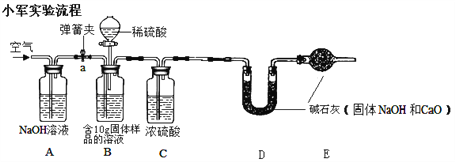
�ټ���װ�������ԣ�װ��ҩƷ�����Ӳ��̶���װ��
�ڴ��ɼ�a��ͨ��һ��ʱ��Ŀ������ٳ���Dװ��������¼Ϊ100g
�۹رյ��ɼ�a����Bװ���еķ�Һ©��������ϡ���ᣬֱ��ƿ�������ݲ�����
�ܴ��ɼ�a�� ����ͨ��һ��ʱ��������ٳ���Dװ��������¼Ϊ102.2g
ʵ�������
��1����С��ʵ����Aװ��������__________
��2��ʵ�鲽����У�����ͨ��һ��ʱ�������Ŀ����_________________
��3�����ȱװ��C,����������������Ƶĺ�������________���ƫ�ߡ��������䡱 �����ߡ�ƫ�͡���
���𰸡� 2NaOH + CO2 = Na2CO3 + H2O ϡ���ᣨϡ���ᣩ �������ƹ����ѱ��� ��Һ���ɫ ������ Na2CO3��Ca(OH)2��CaCO3����2NaOH 47% ��ȥ�����е�CO2����ֹ�䱻D���� ����Ӧ���ɵ�CO2ȫ���ų���D���� ƫ��
���������û�ѧ����ʽ��ʾ���������ڿ����б���ԭ����2NaOH+CO2=Na2CO3+H2O����һƿ�������ƹ���ɷֽ��е�̽����̽��һ��ȷ��һƿ�������ƹ����Ƿ���ʡ�
ʵ����� | ʵ������ | ���� |
ȡ������Ʒ���Թ��У�����һ����ˮ����Ʒȫ�ܽ�μ�������ϡ���ᣨϡ���ᣩ�� | �����ݲ�����ϡ���ᣨϡ���ᣩ��̼���Ʒ�Ӧ���ɶ�����̼���塣 | �������ƹ����ѱ��ʡ� |
̽������ȷ���������ƹ�����ʵij̶ȡ�
ʵ����� | ʵ������ | ���� |
��ȡ������Ʒ���Թ��У�����һ����ˮ����Ʒȫ�ܽ�μӹ������Ȼ�����Һ | ������ɫ������̼�������Ȼ�����Һ��Ӧ�����Ȼ��ƺ�̼��ư�ɫ������ | �������ƹ��岿�� ���� |
�ھ��ú�ȡ�ϲ���Һ���Թ��У�����2�η�̪��Һ | ��Һ���ɫ��˵����Һ��ʱ�ʼ��ԡ� |
������������Һ�����Ȼ�����Һ���÷��������У����ɣ�̼�������������Ʒ�Ӧ����̼��ư�ɫ�������������ơ�̽�����������������ֱ��ʵ��������ƹ������������Ƶ�������������1���������Ʒ��̼��������Ϊx
Na2CO3��CaCl2��CaCO3����2NaCl
106 100
X 5.0g
![]() ��
��![]() ,x��5.3g
,x��5.3g
��Ʒ���������Ƶ�����������![]() ��100����47%��
��100����47%��
�����Ʒ���������Ƶ�����������47%��
��2����ʵ�鲽�����¡�ʵ���������1��ʵ����Aװ�������dz�ȥ�����е�CO2����ֹ�䱻D���գ������к��ж�����̼����2��ʵ�鲽����У�����ͨ��һ��ʱ�������Ŀ���ǽ���Ӧ���ɵ�CO2ȫ���ų���D���ա���3��Ũ���������ˮ�ԣ����ȱװ��C����ö�����̼������ʵ�ʸߣ�̼���ƺ�����ʵ�ʸߣ�����������������Ƶĺ�������ƫ�͡�
�㾦������һ��ʵ��̽���⣬̽�����������ڿ����б���������������Ʊ����ܷⱣ�棬��Ϊ��������ж�����̼������ѧ��Ӧ�����ʡ�

 Сѧ��ʱ��ҵȫͨ����ϵ�д�
Сѧ��ʱ��ҵȫͨ����ϵ�д� �����ÿ�ʱѵ��ϵ�д�
�����ÿ�ʱѵ��ϵ�д� ��Ԫȫ��������ϵ�д�
��Ԫȫ��������ϵ�д� �»ƸԱ����ܾ�ϵ�д�
�»ƸԱ����ܾ�ϵ�д�����Ŀ��̼��þˮ�������Ʊ�þ��Ʒ���м��塣
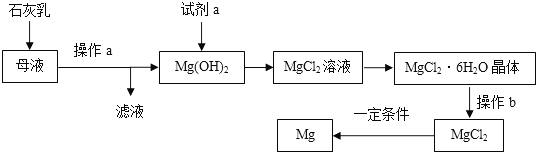
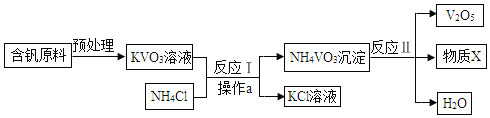
����ȡMgCO33H2O����ҵ�ϴ�������±ˮ(��Ҫ�ɷ�ΪMgCl2)�л�ȡMgCO33H2O�ķ�����ͼ1��
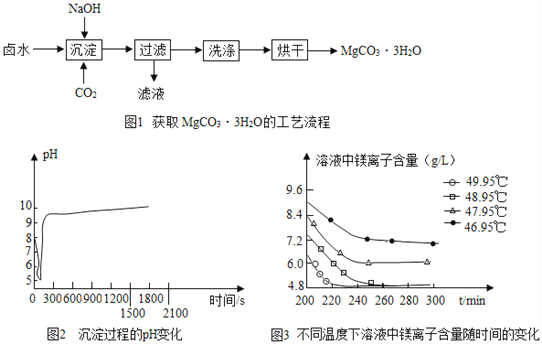
(1)�������̵Ļ�ѧ����ʽΪ��MgCl2+CO2+2NaOH+2H2O�TMgCO33H2O��+2_____��
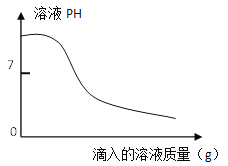
(2)�������̵�pH��ʱ��ı仯��ͼ2��ʾ���������̵IJ���Ϊ_______(����)��
a����±ˮ�еμ�NaOH��Һ��ͬʱͨ��CO2
b����NaOH��Һ�еμ�±ˮ��ͬʱͨ��CO2
c����±ˮ��ͨ��CO2�����ͣ�Ȼ��μ�NaOH��Һ��ͬʱ����ͨ��CO2
d����NaOH��Һ��ͨ��CO2�����ͣ�Ȼ��μ�±ˮ��ͬʱ����ͨ��CO2
(3)�������̵���Һ��þ���Ӻ�����ʱ��ı仯��ͼ3��ʾ����ͬ�¶������õ������������±���ʾ��
�¶�(��) | ���� |
46.95 | MgCO33H2O |
47.95 | MgCO33H2O |
48.95 | MgCO33H2O |
49.95 | Mg5(OH)2(CO3)44H2O |
���������ѡ����¶�Ϊ__________�������Ǵ��¶��£�Mg2+���������ʽϿ죬����Ч�ʽϸߣ��Ҳ�����������������
���ⶨMgCO33H2O�Ĵ��ȡ�
���������ϡ�
(1)��ʯ����CaO��NaOH�Ĺ������
(2)MgCO33H2O+H2SO4=MgSO4+CO2��+4H2O��
(3)Mg5(OH)2(CO3)44H2OҲ���Ա�ʾΪMg(OH)24MgCO34H2O������Է�������Ϊ466���������ᷴӦ����CO2��
��ʵ�鲽�衿
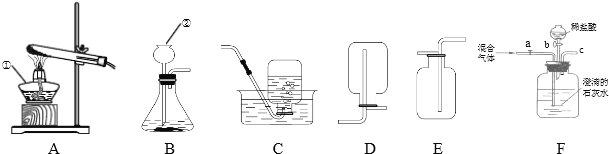
��������ͼ��ʾװ��(ͼ�мг�������ȥ)����ʵ�飬��ȷ��MgCO33H2O�Ĵ��ȡ�
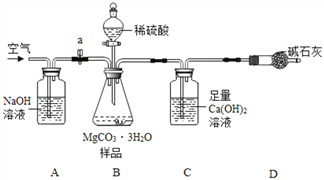
����ʵ����̻ش��������⣺
(1)ʵ����������������ͨ�������������Ϊ______________________��
(2)Cװ���з�Ӧ����CaCO3�Ļ�ѧ����ʽΪ________��D�м�ʯ�ҵ�����Ϊ______________��
(3)���и����ʩ�У�������߲ⶨȷ�ȵ���_______(����)��
a���ڼ�������֮ǰ��Ӧ�ž�װ���ڵ�CO2����
b��Ϊ������ʵ��ʱ�䣬���ٵμ�����
c����A��B֮������ʢ��Ũ�����ϴ��װ��
d����Cװ����ർ��ĩ�������������
(4)С����ΪӦ��Cװ���г���ʯ��ˮ����Ba(OH)2��Һ������һ��____________��ʹCO2�����յĸ���ȫ�����ɶ��� BaCO3����Է���������CaCO3��������MgCO33H2O���ɵ�BaCO3��������CaCO3������ʵ��ʱ������С��
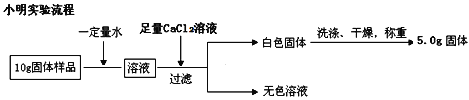
(5)ʵ����ȷ��ȡ15.0g��Ʒ���ݣ��������βⶨ���������CaCO3������ƽ������Ϊ10.0g���������Ʒ��MgCO33H2O�Ĵ���Ϊ___________(д���������)��
(6)����ȡMgCO33H2O����Ʒ�к�������Mg5(OH)2(CO3)44H2O������Ʒ��MgCO33H2O�Ĵ���_________(����ƫ����������������ƫС��)��