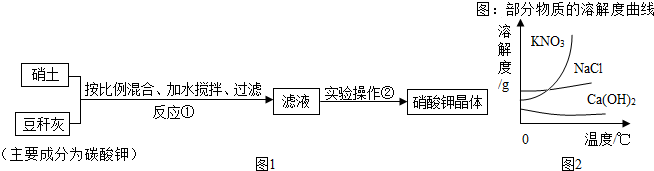
��Ŀ����
������ѧ��ѧ֪ʶ��������ԭ������д��ѧ����ʽ����д����ѧ����ʽ
��1�����ʯ��ʯī���������ʲ����ԭ���� ��
ˮ��˫��ˮ�Ļ�ѧ���ʲ�ͬ��ԭ���� ��
��2����ʢʯ��ˮ���Լ�ƿƿ�ڳ���һ���ɫ��Ĥ����ԭ���� ��
��3���������������⣬��ԭ���� ��
��4������ɭ�ֻ��ֵ���Ч����֮һ���ǽ��������·��ǰ��һƬ��ľ�����������ԭ���� ��
��5����¯�������Դ�����Ϊ���������е���Ҫ��Ӧԭ���� ��
��6��̼�������׳�С�մ�һЩ����С�մ�ı��ɾ��л������dz�˵��θ����֢״�����ã���ԭ���� ��
��1�����ʯ��ʯī���������ʲ����ԭ����
ˮ��˫��ˮ�Ļ�ѧ���ʲ�ͬ��ԭ����
��2����ʢʯ��ˮ���Լ�ƿƿ�ڳ���һ���ɫ��Ĥ����ԭ����
��3���������������⣬��ԭ����
��4������ɭ�ֻ��ֵ���Ч����֮һ���ǽ��������·��ǰ��һƬ��ľ�����������ԭ����
��5����¯�������Դ�����Ϊ���������е���Ҫ��Ӧԭ����
��6��̼�������׳�С�մ�һЩ����С�մ�ı��ɾ��л������dz�˵��θ����֢״�����ã���ԭ����
���㣺̼���ʵ��������ʼ���;,���÷�����ԭ�ӵ����ʷ����ͽ������,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ,����ԭ���ͷ���
ר�⣺���ʵ���������;,��ѧ����������غ㶨��
��������1�����ʵĽṹ���������ʵ����ʣ�
��2��ʯ��ˮ��Ca��OH��2��Һ����������еĶ�����̼������Ӧ���ɰ�ɫ��������ɫ���壩̼��ƣ�
��3���������ڿ��������γ����ܵ�����������Ĥ���������������ԭ��
��4�����ķ����У���1�������������������������2�����µ���ȼ����Ż�����£���3�����߿�ȼ�
��5���ô����������ķ�Ӧԭ�����ڸ��������£����ý�̿��������Ӧ���ɵ�һ����̼����ԭ����������ʯ�н�����ԭ������
��6�������˵�θ�����Ҫ�ɷ������ᣬ�������̼�����Ʒ�Ӧ���з�����
��2��ʯ��ˮ��Ca��OH��2��Һ����������еĶ�����̼������Ӧ���ɰ�ɫ��������ɫ���壩̼��ƣ�
��3���������ڿ��������γ����ܵ�����������Ĥ���������������ԭ��
��4�����ķ����У���1�������������������������2�����µ���ȼ����Ż�����£���3�����߿�ȼ�
��5���ô����������ķ�Ӧԭ�����ڸ��������£����ý�̿��������Ӧ���ɵ�һ����̼����ԭ����������ʯ�н�����ԭ������
��6�������˵�θ�����Ҫ�ɷ������ᣬ�������̼�����Ʒ�Ӧ���з�����
����⣺��1����Ϊ���ʯ��ʯī���ڲ�̼ԭ�ӵ����з�ʽ��ͬ���������������������ʲ���ܴ�ˮ��˫��ˮ��Ԫ�������ͬ���������ʻ�ѧ���ʲ�ͬ������Ϊ���Ƿֱ�����ˮ���ӡ�˫��ˮ���ӹ��ɵģ���ͬ�����ʵķ������ʲ�ͬ��
��2����������������Һ��ʯ��ˮ���Ϳ����еĶ�����̼��Ӧ���ɰ�ɫ����̼��ƣ�CaCO3����ˮ����˾��ÿ�����ʯ��ˮƿ���Ͼͻ���һ���ɫ���壬����ɫ������Ҫ�ɷ־���̼��ƣ���Ӧ�ķ���ʽ�ǣ�Ca��OH��2+CO2=CaCO3��+H2O��
��3���������ڿ���������������Ӧ�������ܵ�����������Ĥ���Ӷ���ֹ����һ�������������Խ������������⣻
��4�����������·��ǰ��һƬ��ľ������Ϊ�˽���ȼ�����Դ���룮
��5���ô����������ķ�Ӧԭ�����ڸ��������£����ý�̿��������Ӧ���ɵ�һ����̼����ԭ����������ʯ�н�����ԭ��������Ӧ����ʽΪ��Fe3O4+4CO
3Fe+4CO2 ��
��6��̼�����ƺ����ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��
�ʴ�Ϊ����1��̼ԭ�ӵ����з�ʽ��ͬ�����߷��ӹ��ɲ�ͬ��
��2��CO2+Ca��OH��2=CaCO3��+H2O��
��3��4Al+3O2=2Al2O3��
��4�������ȼ�
��5��Fe3O4+4CO
3Fe+4CO2��
��6��NaHCO3+HCl=NaCl+H2O+CO2����
��2����������������Һ��ʯ��ˮ���Ϳ����еĶ�����̼��Ӧ���ɰ�ɫ����̼��ƣ�CaCO3����ˮ����˾��ÿ�����ʯ��ˮƿ���Ͼͻ���һ���ɫ���壬����ɫ������Ҫ�ɷ־���̼��ƣ���Ӧ�ķ���ʽ�ǣ�Ca��OH��2+CO2=CaCO3��+H2O��
��3���������ڿ���������������Ӧ�������ܵ�����������Ĥ���Ӷ���ֹ����һ�������������Խ������������⣻
��4�����������·��ǰ��һƬ��ľ������Ϊ�˽���ȼ�����Դ���룮
��5���ô����������ķ�Ӧԭ�����ڸ��������£����ý�̿��������Ӧ���ɵ�һ����̼����ԭ����������ʯ�н�����ԭ��������Ӧ����ʽΪ��Fe3O4+4CO
| ||
��6��̼�����ƺ����ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��
�ʴ�Ϊ����1��̼ԭ�ӵ����з�ʽ��ͬ�����߷��ӹ��ɲ�ͬ��
��2��CO2+Ca��OH��2=CaCO3��+H2O��
��3��4Al+3O2=2Al2O3��
��4�������ȼ�
��5��Fe3O4+4CO
| ||
��6��NaHCO3+HCl=NaCl+H2O+CO2����
������������Ҫ�������ʵ����ʺ���;�����ʱҪ�������������ʵ����ʣ�Ȼ���ٸ������ʵ����ʷ�����з������жϣ��Ӷ�ȷ�����ʵ���;���������˻�ѧ����ʽ����д������

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
����ʩ�ã�NH4��2SO4���ʵ����������ữ������˵������ȷ���ǣ�������
| A����NH4��2SO4���ڵ��� |
| B����NH4��2SO4������ˮ |
| C�����˳���ʩ�ã�NH4��2SO4���� |
| D����NH4��2SO4����������ʯ�һ��ʩ�� |
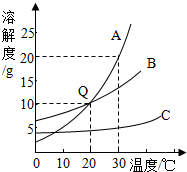 ��ͼΪA��B��C���ֹ��������ܽ�����ߣ�
��ͼΪA��B��C���ֹ��������ܽ�����ߣ�