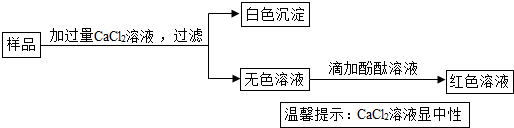
��Ŀ����
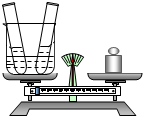 ij��ȤС��Ϊ����֤�����غ㶨�ɣ�������С�Թ��зֱ�ע��20g������������Ϊ20%������������Һ��20g����ͭ��Һ��Һ������һ�����ձ��г�������ͼ5��ʾ��Ȼ����ֻС�Թ���ҩƷ�����ձ��У��Թܲ�ȡ������ǡ����ȫ��Ӧ���Ը���Ҫ�ش��������⣺
ij��ȤС��Ϊ����֤�����غ㶨�ɣ�������С�Թ��зֱ�ע��20g������������Ϊ20%������������Һ��20g����ͭ��Һ��Һ������һ�����ձ��г�������ͼ5��ʾ��Ȼ����ֻС�Թ���ҩƷ�����ձ��У��Թܲ�ȡ������ǡ����ȫ��Ӧ���Ը���Ҫ�ش��������⣺��1��д��ʵ������й۲쵽��һ������
��2����Ӧ��������ƽָ���
��������ƫת��֮һ����
��3���Լ���ǡ����ȫ��Ӧʱ���ò�������Һ���������������ȷ��0.1g��
���㣺�����غ㶨�ɼ���Ӧ��,���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺�йػ�ѧ����ʽ�ļ���,��ѧ����������غ㶨��
�����������������ƺ�����ͭ��Ӧ������������ͭ����ɫ��������������ͭ�ܹ������ᷴӦ��������ͭ��ˮ������Ӧ�������������غ㶨�ɵ��й�֪ʶ�ж������ı仯�����÷���ʽ�Ļ������㼴�ɼ������Ӧ������Һ��������
����⣺��1�������������ƺ�����ͭ��Ӧ�ܹ�����������ͭ����ɫ�����������ƣ���������ͭ�ܹ���ϡ���ᷴӦ��������ͭ��ˮ�����������ǣ�������ɫ���������������ɫ������
��2�����ڷ�Ӧǰ��û�����ʴ�ʵ��װ�����ܳ����������������غ㶨�ɿ�֪װ�õ���������仯������ƽָ�벻��ƫת�����������ƫת��
��3����Ϊ����ǡ����ȫ��Ӧ����20g20%������������Һ������ͭ��ȫ��Ӧ������������ͭ����������Ϊx����
2NaOH+CuSO4�TNa2SO4+Cu��OH��2��
80 98
20g��20% x
=
x=4.9g
���������غ㶨�ɿ�֪����Ӧ�����ò�������Һ������Ϊ��20g+20g-4.9g=35.1g��
��2�����ڷ�Ӧǰ��û�����ʴ�ʵ��װ�����ܳ����������������غ㶨�ɿ�֪װ�õ���������仯������ƽָ�벻��ƫת�����������ƫת��
��3����Ϊ����ǡ����ȫ��Ӧ����20g20%������������Һ������ͭ��ȫ��Ӧ������������ͭ����������Ϊx����
2NaOH+CuSO4�TNa2SO4+Cu��OH��2��
80 98
20g��20% x
| 80 |
| 98 |
| 20g��20% |
| x |
x=4.9g
���������غ㶨�ɿ�֪����Ӧ�����ò�������Һ������Ϊ��20g+20g-4.9g=35.1g��
���������⿼���������غ㶨�ɵ�Ӧ�ã���ɴ��⣬�����������е�֪ʶ�ͻ�ѧ����ʽ�Ļ������㼴�ɣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
���෨�ǻ�ѧѧϰ���о�����Ҫ����֮һ�����з�����ȷ���ǣ�������
| A���ǽ���Ԫ�أ�Mg��F��Si |
| B�������ˮ��������̼������þ |
| C�����ʣ�������Һ����ˮ |
| D������������ʯ�͡���ˮ����� |




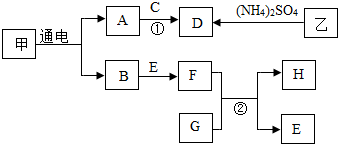 ��ͼ��ʾ�ļ��г�������ת����ϵ��A��H���ס��ҷֱ��ʾ��ͬ���ʣ�����A��B��C��H��Ϊ��ɫ��ζ���壬DΪ�̼�����ζ���壬EΪ������������Ӧ�������Ľ������ʣ��ҿ������к��������ԣ���Ӧ��Ϊ���Ϸ�Ӧ�������ַ�Ӧ������P��Ӧ��������ȥ������ش��������⣺
��ͼ��ʾ�ļ��г�������ת����ϵ��A��H���ס��ҷֱ��ʾ��ͬ���ʣ�����A��B��C��H��Ϊ��ɫ��ζ���壬DΪ�̼�����ζ���壬EΪ������������Ӧ�������Ľ������ʣ��ҿ������к��������ԣ���Ӧ��Ϊ���Ϸ�Ӧ�������ַ�Ӧ������P��Ӧ��������ȥ������ش��������⣺