��Ŀ����
�������Ź㷺��Ӧ�ã�
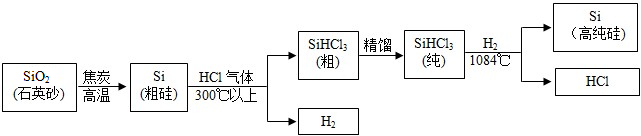
��1�����н�����Ʒ�У����ý��������Ե���______������ĸ��ţ���
��2����ҵ����һ����̼�ͳ�������Ҫ�ɷ����������������Ļ�ѧ����ʽΪ______��
��3��������������������������У���Һ����ɫ��Ϊ��ɫ����������ð����д���йط�Ӧ�Ļ�ѧ����ʽ______��
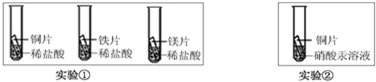
��4��ij���������ŷŵķ�Һ�к�������������������ͭ��Ϊ����������Դ�ͷ�ֹ��Ⱦ���ó����Һ�м���һ���������ۣ���Ӧֹͣ����ˣ����˳��Ĺ����м���ϡ���ᣬ������������������й�˵���У���ȷ����______�� ������ĸ��ţ���
A���˳��Ĺ�����һ����������һ��������
B���˳��Ĺ�����һ�������������ܺ���ͭ����
C����Һ��һ��������������������������������ͭ
D����Һ��һ��������������һ��û��������������ͭ��
�⣺��1�����������ý����ĵ����ԣ����������ý����ĵ����ԣ����������ý�������չ�ԣ���ѡB��
��2����Ӧ����һ����̼�������������������Ͷ�����̼���õ�ʧ��������ƽ����Ӧ�����Ǹ��£����Է���ʽ�ǣ�3CO+Fe2O3 2Fe+3CO2��
2Fe+3CO2��
��3������������������Ӧ�����Ȼ�����ˮ����Ӧ��������������������������Ȼ�����ˮ���ù۲취��ƽ�����Է���ʽ�ǣ�6HCI+Fe2O3�T2FeCI3+3H2O�������ⷴӦ�꣬��������Ӧ�����Ȼ��������������ù۲취��ƽ��������������������ţ����Է���ʽ�ǣ�Fe+2HCI�TFeCI2+H2����
��4���������֪�����˳��Ĺ����м�������ϡ���ᣬû�����������˵��������û��������������������Ӧ����������ͭ��Ӧ���˳��Ĺ�����һ������������ͭ��һ��û����
A���˳��Ĺ�����һ�������������ܺ���ͭ��һ������������A��ȷ��
B���˳��Ĺ�����һ�������������ܺ���ͭ��һ������������B����
C�����˳��Ĺ����м�������ϡ���ᣬû�����������˵��������û��������������������Ӧ����������ͭ��Ӧ����Һ��һ��������������������������������ͭ����C��ȷ��
D����Һ��һ��������������������������������ͭ����D����
��ѡAC��
�ʴ�Ϊ����1��B����2��3CO+Fe2O3 2Fe+3CO2����3��6HCI+Fe2O3�T2FeCI3+3H2O��Fe+2HCI�TFeCI2+H2������4��AC��
2Fe+3CO2����3��6HCI+Fe2O3�T2FeCI3+3H2O��Fe+2HCI�TFeCI2+H2������4��AC��
��������1�����ݽ��������ʾ�����������;����������;��ӳ���������ʿ��ǣ���2�����ݷ���ʽ����д�������ǣ���3������������������Ӧ�����Ȼ�����ˮ�������ⷴӦ�꣬��������Ӧ�����Ȼ���������������4���ڽ������˳���У�������ͭ������ǰ�ߣ����Ժ�������������ͭ��Һ��Ӧ����������ͭ�Լ��������������˳��Ĺ����м�������ϡ���ᣬ�����������˵��ʣ��Ĺ�����û������
���������⿼��Ľ������������ʣ�������������Һ�ķ�Ӧ����ѧ����ʽ����д����ɴ��⣬�������ݽ������˳���Լ�����ṩ����Ϣ���У�����Ҫ��ͬѧ����ƽʱ��ѧϰ�м�ǿ����֪ʶ�ļ��䣬�Ա��ܹ����Ӧ�ã�
��2����Ӧ����һ����̼�������������������Ͷ�����̼���õ�ʧ��������ƽ����Ӧ�����Ǹ��£����Է���ʽ�ǣ�3CO+Fe2O3
 2Fe+3CO2��
2Fe+3CO2����3������������������Ӧ�����Ȼ�����ˮ����Ӧ��������������������������Ȼ�����ˮ���ù۲취��ƽ�����Է���ʽ�ǣ�6HCI+Fe2O3�T2FeCI3+3H2O�������ⷴӦ�꣬��������Ӧ�����Ȼ��������������ù۲취��ƽ��������������������ţ����Է���ʽ�ǣ�Fe+2HCI�TFeCI2+H2����
��4���������֪�����˳��Ĺ����м�������ϡ���ᣬû�����������˵��������û��������������������Ӧ����������ͭ��Ӧ���˳��Ĺ�����һ������������ͭ��һ��û����
A���˳��Ĺ�����һ�������������ܺ���ͭ��һ������������A��ȷ��
B���˳��Ĺ�����һ�������������ܺ���ͭ��һ������������B����
C�����˳��Ĺ����м�������ϡ���ᣬû�����������˵��������û��������������������Ӧ����������ͭ��Ӧ����Һ��һ��������������������������������ͭ����C��ȷ��
D����Һ��һ��������������������������������ͭ����D����
��ѡAC��
�ʴ�Ϊ����1��B����2��3CO+Fe2O3
 2Fe+3CO2����3��6HCI+Fe2O3�T2FeCI3+3H2O��Fe+2HCI�TFeCI2+H2������4��AC��
2Fe+3CO2����3��6HCI+Fe2O3�T2FeCI3+3H2O��Fe+2HCI�TFeCI2+H2������4��AC����������1�����ݽ��������ʾ�����������;����������;��ӳ���������ʿ��ǣ���2�����ݷ���ʽ����д�������ǣ���3������������������Ӧ�����Ȼ�����ˮ�������ⷴӦ�꣬��������Ӧ�����Ȼ���������������4���ڽ������˳���У�������ͭ������ǰ�ߣ����Ժ�������������ͭ��Һ��Ӧ����������ͭ�Լ��������������˳��Ĺ����м�������ϡ���ᣬ�����������˵��ʣ��Ĺ�����û������
���������⿼��Ľ������������ʣ�������������Һ�ķ�Ӧ����ѧ����ʽ����д����ɴ��⣬�������ݽ������˳���Լ�����ṩ����Ϣ���У�����Ҫ��ͬѧ����ƽʱ��ѧϰ�м�ǿ����֪ʶ�ļ��䣬�Ա��ܹ����Ӧ�ã�

��ϰ��ϵ�д�
�����Ŀ