��Ŀ����
ijУѧϰС����к��������Ƿ�����ЧԤ��ȣ�ݵ�ʵ��̽����
��Ϣ���ٵ�����Ҫ�ɷ���CaCO3�������ɷֶ�ʵ��Ӱ����Բ��ơ�
�ڼ����������ݺͺ������෴Ӧ�Ļ�ѧԭ�����ơ�
��ʵ��һ����1�������ʵļ�������ʢ������ϡ������ձ��У��ɹ۲쵽��������������ݣ�����������ݳɷֵķ�����_____���û�ѧ����ʽ��ʾ����
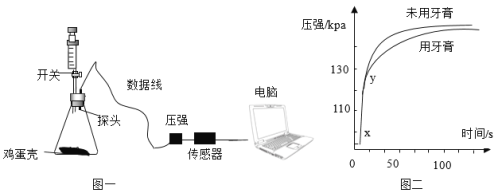
��ʵ����������Ǵ�������̽�����������ܷ�Ԥ��ȣ�ݡ�
������֪���������ࣨ�����ɷ�ΪNaF�����뵰�Ƿ�Ӧ���ڱ������ɷ����Ʊ����㡣��Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2NaF==CaF2+Na2CO3
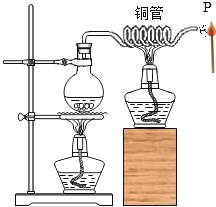
��2����ͼһ���Ӻ�װ�ã��ȼ��װ�������ԣ�������_____���ٳ�ȡ0.50g�����ദ�����ĵ��ǣ�������Һ���ݡ�ϴ�ӡ����ɣ�������ƿ�У���ע������ȡ20mLϡ���ᣨģ���γ�ȣ�ݵ��ᾳ�������Ӻ�װ�ã����أ����������ѹ����ƿ���رտ��أ����ѹǿ��ʱ��ı仯��ͼ����ʾ���á�δ�����ദ���ĵ��ǡ��ظ�����ʵ�顣��ע�������Ǽ���Һ����ɵ�ѹǿ�
��3��ͼ����x-y��ѹǿ�������Ҫԭ��_____���á�δ�����ദ���ĵ��ǡ��ظ�����ʵ���������_____��
��4������ʵ��������ݷ�������������_____����ܡ����ܡ���Ԥ��ȣ�ݣ�����_____��
�±���С��ͬѧ�ö��ַ����������ʵ�������������÷�����ȫ��ȷ����
��Ҫ��������� | �����������Լ� | ||
����һ | ������ | ||
A | �Ȼ�����Һ���Ȼ�ͭ��Һ | �۲���ɫ | �μ���������Һ |
B | ̼���ƺ��Ȼ��� | ��������ˮ�� | �������� |
C | �ռ���Һ�ͳ���ʯ��ˮ | �۲���ɫ | ͨ�������̼���� |
D | Ũ�����Ũ���� | ��ƿ�ڹ۲� | ����ζ |
A.A B.B C.C D.D


 ��ȩ B.
��ȩ B. �ƾ�
�ƾ� �ռ� D.
�ռ� D. Ũ����
Ũ����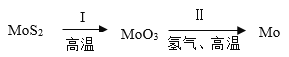
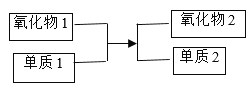
 2MoO3+4_____
2MoO3+4_____