��Ŀ����
����Ŀ���˵������벻����ѧ�����û�ѧ֪ʶ�ش���������
��1���ಽ�У��ﵥ�����У�����Ч����CO2��SO2��CO��������ŷţ���Щ����������������ЧӦ����_____��������������_____��
��2����ʯȼ�ϰ���ú����Ȼ����_____����������_____�������������������������Դ��������Ȼ������Ҫ�ɷ�Ϊ��_____����Ȼ���ڿ�������ȫȼ�յĻ�ѧ����ʽΪ��_____��
��3��ʹȼ�ϳ��ȼ�ն��ڽ�Լ��Դ�dz���Ҫ��ȼ�ϳ��ȼ��ͨ���������㣺һ��ȼ��ʱҪ���㹻�Ŀ���������_____������Դ�����ÿ��Բ��ֽ����ʯ��Դ���ٺľ������⣬Ŀǰ�������ڿ��������õ�����Դ��_____����һ�֣���
��4������ʱ���е��Ͳ����Ż𣬿��ù��Ǹ��������ԭ��Ϊ��_____��
���𰸡�CO2 SO2 ʯ�� �������� ���� CH4+2O2 CO2+2H2O ȼ�������Ҫ���㹻��ĽӴ���� ̫���� ��������
CO2+2H2O ȼ�������Ҫ���㹻��ĽӴ���� ̫���� ��������
��������
(1)������̼����������ЧӦ�����������������꣬���Զಽ�У��ﵥ�����У�����Ч����CO2��SO2��CO��������ŷţ�����д��CO2��SO2��
(2)��ʯȼ�ϰ���ú����Ȼ����ʯ�ͣ��������ڲ���������Դ������д��ʯ�ͣ�����������
��Ȼ������Ҫ�ɷ��Ǽ��飬����д�����飻
��Ȼ���ڿ�������ȫȼ���Ǽ����������������Ӧ����ˮ�Ͷ�����̼������д��CH4+2O2![]() CO2+2H2O��
CO2+2H2O��
(3)ʹȼ�ϳ��ȼ��ͨ���������㣺һ��ȼ��ʱҪ���㹻�Ŀ���������ȼ�������Ҫ���㹻��ĽӴ����������д��ȼ�������Ҫ���㹻��ĽӴ������
Ŀǰ�������ڿ��������õ�����Դ��̫���ܡ����ܡ����ܡ������ܡ������ܵȣ�����д��̫���ܣ��𰸲�Ψһ����
(4)����ʱ���е��Ͳ����Ż𣬿��ù��Ǹ��������ԭ��Ϊ����������������д������������

 �㾦�½̲�ȫ�ܽ��ϵ�д�
�㾦�½̲�ȫ�ܽ��ϵ�д� Сѧ�̲���ȫ���ϵ�д�
Сѧ�̲���ȫ���ϵ�д�����Ŀ������һ����°����ҹ������Ļ�ѧ�ң������˺����Ƽ��Ϊ����͵��ʹ�ҵ�����ķ�չ�����˽ܳ��Ĺ��ס�������ʵ����ģ������Ƽԭ�����������̣�

��20��ʱ����CO2ͨ�뺬NH3�ı���NaCl��Һ�У����� NaHCO3�����NH4Cl��Һ��
�������еIJ��� NaHCO3�����ּ��ȷֽ⣬�õ�Na2CO3��H2O��CO2��
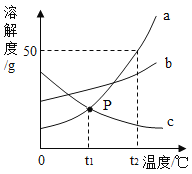
���϶���20��ʱ����������ˮ�е��ܽ��/g
NaCl | NH4Cl | NaHCO3 |
36.0 | 37.2 | 9.6 |
��ش��������⣺
��1���ڵ�������Ӧ�У��� NaHCO3������������û��NH4Cl��NaCl����������ԭ����_____��
��2�������������в������������Ϊԭ��ѭ��ʹ�õ���_____��ͬʱ�õ��ĸ���ƷNH4Cl��һ�ֵ��ʡ�