��Ŀ����
ij��ѧ��ȤС����Ƴ����д��η����ᴿ��ʵ�鷽��������һ��������������⣮
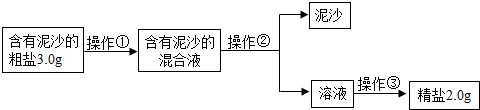
��1�������٢ڢ۵�����������
��2��ʵʩ������ʱ������Ҫ���Ͻ��裬ֱ��
��3�����㾫�εIJ���ƫ��
��4�������εIJ���ƫ�ͣ�����ܵ�ԭ����
A������ʱ��ֽ������
B������ʱҺ���н���
C���ܽ⺬����ɳ�Ĵ���ʱ�������ˮ������
D��ת�ƾ���ʱ���������в����϶����
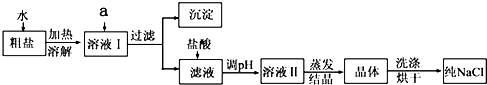
��5�������л�����CaCl2��Na2SO4�����ʣ�Ϊ�˳�ȥ���������ʣ����μ��������BaCl2��Һ��Na2CO3��Һ�����ˣ�������Һ�м������������ᣮʵ���м��������BaCl2��Һ��Ϊ�˳�ȥ
��1�������٢ڢ۵�����������
�ܽ�
�ܽ�
������
����
������
����
��2��ʵʩ������ʱ������Ҫ���Ͻ��裬ֱ��
���ִ�������
���ִ�������
ʱֹͣ���ȣ���3�����㾫�εIJ���ƫ��
66.7%
66.7%
������������һλС��������4�������εIJ���ƫ�ͣ�����ܵ�ԭ����
A������ʱ��ֽ������
B������ʱҺ���н���
C���ܽ⺬����ɳ�Ĵ���ʱ�������ˮ������
D��ת�ƾ���ʱ���������в����϶����
��5�������л�����CaCl2��Na2SO4�����ʣ�Ϊ�˳�ȥ���������ʣ����μ��������BaCl2��Һ��Na2CO3��Һ�����ˣ�������Һ�м������������ᣮʵ���м��������BaCl2��Һ��Ϊ�˳�ȥ
SO42-
SO42-
�����������Na2CO3��Һ��Ŀ������ȫ��ȥ��Һ�е�Ca2+��Ba2+
��ȫ��ȥ��Һ�е�Ca2+��Ba2+
��
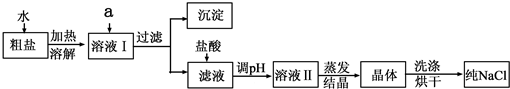
��������1�����ݴ����ᴿ�Ļ������裺�ܽ⡢���ˡ�����������
��2�����������ᾧ�����ִ��������ֹͣ�����������Ƚ�ʣ��ˮ������������
��3���þ��ε��������Դ��ε��������
��4������A����ʱ��ֽ���������ʳ�ε���ʧ��B����ʱҺ���н���ʳ�β���ȫ���ᾧ������C�ܽ⺬����ɳ�Ĵ���ʱ�������ˮ��������Һ�е�ʳ�ε���������ԭ����ô�ࡢDת�ƾ���ʱ�������в����϶���壬ʹ�����е�ʳ����ʧһ���ַ�����
��5������Ba2+��SO42-��ϳ����ᱵ����������̼��������ӡ������Ӷ��ܷ�Ӧ�ֱ�����̼��ƺ�̼�ᱵ���ֳ���������
��2�����������ᾧ�����ִ��������ֹͣ�����������Ƚ�ʣ��ˮ������������
��3���þ��ε��������Դ��ε��������
��4������A����ʱ��ֽ���������ʳ�ε���ʧ��B����ʱҺ���н���ʳ�β���ȫ���ᾧ������C�ܽ⺬����ɳ�Ĵ���ʱ�������ˮ��������Һ�е�ʳ�ε���������ԭ����ô�ࡢDת�ƾ���ʱ�������в����϶���壬ʹ�����е�ʳ����ʧһ���ַ�����
��5������Ba2+��SO42-��ϳ����ᱵ����������̼��������ӡ������Ӷ��ܷ�Ӧ�ֱ�����̼��ƺ�̼�ᱵ���ֳ���������
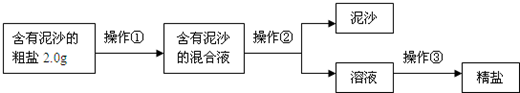
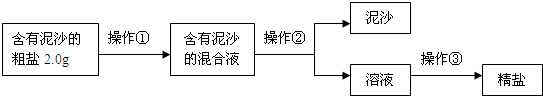
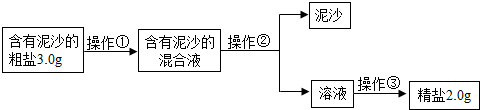
����⣺��1�������ᴿ�Ļ������裺�ܽ⡢���ˡ��������ʴ𰸣��ܽ⡢���ˡ������ᾧ��
��2�������ᾧ�����ִ��������ֹͣ�����������Ƚ�ʣ��ˮ���������ʴ𰸣����ִ������壮
��3���þ��ε��������Դ��ε����������ʣ�
��100%=66.7%���ʴ𰸣�66.7%��
��4��A����ʱ��ֽ���������ʳ�ε���ʧ��B����ʱҺ���н���ʳ�β���ȫ���ᾧ������C�ܽ⺬����ɳ�Ĵ���ʱ�������ˮ��������Һ�е�ʳ�ε���������ԭ����ô�ࡢDת�ƾ���ʱ�������в����϶���壬ʹ�����е�ʳ����ʧһ���֣��ʴ𰸣�BD��
��5��Ba2+��SO42-��ϳ����ᱵ����������̼��������ӡ������Ӷ��ܷ�Ӧ�ֱ�����̼��ƺ�̼�ᱵ���ֳ������ʴ𰸣�S
����ȫ��ȥ��Һ�е�Ca2+��Ba2+
��2�������ᾧ�����ִ��������ֹͣ�����������Ƚ�ʣ��ˮ���������ʴ𰸣����ִ������壮
��3���þ��ε��������Դ��ε����������ʣ�
| 2g |
| 3g |
��4��A����ʱ��ֽ���������ʳ�ε���ʧ��B����ʱҺ���н���ʳ�β���ȫ���ᾧ������C�ܽ⺬����ɳ�Ĵ���ʱ�������ˮ��������Һ�е�ʳ�ε���������ԭ����ô�ࡢDת�ƾ���ʱ�������в����϶���壬ʹ�����е�ʳ����ʧһ���֣��ʴ𰸣�BD��
��5��Ba2+��SO42-��ϳ����ᱵ����������̼��������ӡ������Ӷ��ܷ�Ӧ�ֱ�����̼��ƺ�̼�ᱵ���ֳ������ʴ𰸣�S
| O | 2- 4 |
���������ֽⷴӦ��ʵ�������Ӽ�ķ�Ӧ���������ӽ�ϳɳ����������ˮ���ܷ�����Ӧ��

��ϰ��ϵ�д�
 ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д�
ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д� ���������ν�ϵ�д�
���������ν�ϵ�д�
�����Ŀ