��Ŀ����
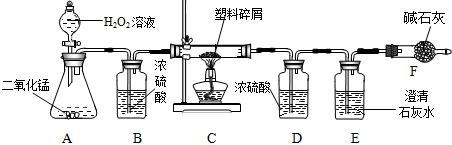
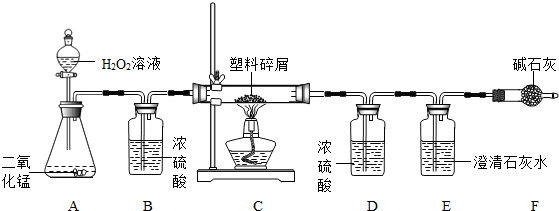
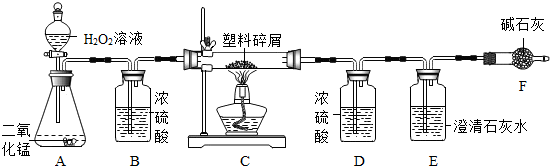
���ڴ���ʹ��һ�������Ϸ��������ɵġ���ɫ��Ⱦ���ѳ�Ϊһ�����ص�������⡣ij��ѧ�о�С���ͬѧ��ij�����ϴ�����ɽ��з����о�(������ ʾ������ֻ��C��H����Ԫ��)�������������ͼ��ʾ��ʵ��װ��,ʹ�����������ڴ�������ȫȼ��,�۲�ʵ���������й����ݡ�����Ԫ�غ�����(��ʯ�ҿ�����ˮ�Ͷ�����̼)
ʾ������ֻ��C��H����Ԫ��)�������������ͼ��ʾ��ʵ��װ��,ʹ�����������ڴ�������ȫȼ��,�۲�ʵ���������й����ݡ�����Ԫ�غ�����(��ʯ�ҿ�����ˮ�Ͷ�����̼)

(1)ʵ��װ������һ�����Դ���,��д���������� �� ��
(2)װ��A�з�Ӧ�Ļ�ѧ����ʽΪ �� ��
(3)װ��E�е��������� ,
װ��F���������� ��
(4)��װ��C�IJ������з����������������Ϊ5.9 g,�����������ȼ�պ�,װ��D����7.2 g,������������к���Ԫ�ص�����Ϊ��������g��
(5)��װ����û������װ��B,��ʹ��������������Ԫ�ص�����������
��������(�ƫС����ƫ����Ӱ�족)��
װ��B��Ũ����������Ǹ�������,����Ӧ�����̳�;A�з����ķ�Ӧ�ǹ��������ڶ������̵Ĵ�����������ˮ������;������м��������ȼ�����ɵĶ�����̼ͨ�����ʯ��ˮ,ʹ����ʯ��ˮ�����,F�м�ʯ�ҵ����������տ����еĶ�����̼,��ֹ�����еĶ�����̼��ˮ����E��ʹ���������ȷ;װ��D����7.2 g,˵��ȼ������ˮ������Ϊ7.2 g,���Ը����������к���Ԫ�ص�����Ϊ0.8 g,B�������Ǹ�������,���û������װ��B,��ʹˮ������ƫ��,��ʹ��������������Ԫ�ص�����������ƫ��
��:(1)B�е���Ӧ�����̳�
(2)2H2O2 2H2O+O2��
2H2O+O2��
(3)����ʯ��ˮ����ǡ���ֹ�����еĶ�����̼��ˮ����E��ʹ���������ȷ
(4)0.8��(5)ƫ��

 Ӯ�ڿ�����ʦ��ʱ�ƻ�ϵ�д�
Ӯ�ڿ�����ʦ��ʱ�ƻ�ϵ�д� �������Ͽ�ʱͬ��ѵ��ϵ�д�
�������Ͽ�ʱͬ��ѵ��ϵ�д�