��Ŀ����
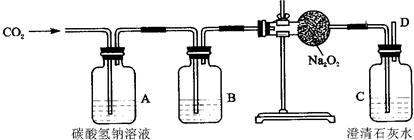
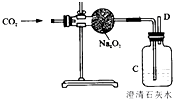 ���϶����š��ɴ��ijɹ��������ҹ�������ҵ����һ��̱���������������ȿ�������ɴ���DZˮͧ�е��������������磺�������ƣ�Na2O2���ڳ����������˺����Ķ�����̼��Ӧ����������̼���ƣ�Ϊ����֤�÷�Ӧ�������IJ�����ij��ȤС���ͬѧ�������ͼ��ʾ��ʵ�飮
���϶����š��ɴ��ijɹ��������ҹ�������ҵ����һ��̱���������������ȿ�������ɴ���DZˮͧ�е��������������磺�������ƣ�Na2O2���ڳ����������˺����Ķ�����̼��Ӧ����������̼���ƣ�Ϊ����֤�÷�Ӧ�������IJ�����ij��ȤС���ͬѧ�������ͼ��ʾ��ʵ�飮��1��Ϊ��ȷ��CO2��Na2O2��Ӧ���������������������Ҫ���õ���֤ʵ���������D���ܴ�����һ��
��2��
��3����ÿ��ÿ������Ķ�����̼������Ϊ2.2ǧ�ˣ����ù������ƽ���Щ������̼ȫ�����գ��������Ͽɲ�����������ǧ�ˣ���д��������̣�
��4����ʵ�����ø�����ؼ���Ҳ�ܲ���������С��ͬѧ��16g�ĸ�����ط����Թ��м���һ��ʱ�����ȴ���Ƶ�ʣ�����ʵ�����Ϊ14.4g��
�ٴ�ҷײ������ù������ʵijɷ֣�д���ʵĻ�ѧʽ����
����һ��
�������
�ھ�������ȷ������
��������1��������������������ɽ��
��2�����ö�����̼��ʹ����ʯ��ˮ����ǵ����ʻش�
��3�����û�ѧ����ʽ���м��㣬д��������̣�
��4�����ݸ���������ȷֽ�Ļ�ѧ����ʽ���ɵ�֪��Ӧ�и��������������������ϵ�����������������������ɼ�������Ҫ������ص�����������Ʋ⣺���������ȫ��Ӧ����ȫ��Ӧ����������룻��������ȷ���ĸ���������ȷ�ģ�
��2�����ö�����̼��ʹ����ʯ��ˮ����ǵ����ʻش�
��3�����û�ѧ����ʽ���м��㣬д��������̣�
��4�����ݸ���������ȷֽ�Ļ�ѧ����ʽ���ɵ�֪��Ӧ�и��������������������ϵ�����������������������ɼ�������Ҫ������ص�����������Ʋ⣺���������ȫ��Ӧ����ȫ��Ӧ����������룻��������ȷ���ĸ���������ȷ�ģ�
����⣺
��1����Ϊ������֧��ȼ�գ��ʿ����ô����ǵ�ľ�����ڼ���ƿ�ڼ���������
��2���������ʯ��ˮû�б仯����˵��������̼�ѱ�����������ȫ���գ�
��3����ÿ��ÿ������Ķ�����̼������Ϊ2.2ǧ�ˣ�����ݻ�ѧ����ʽ���Լ��㣮
�⣺�������Ͽɲ�������������Ϊx��
2Na2O2+2CO2=2Na2CO3+O2
88 32
2.2kg x
��
=
�����x=0.8kg
���������Ͽɲ�������0.8ǧ�ˣ�
��4���ٸ��ݻ�ѧ����ʽ2KMnO4
K2MnO4+MnO2+O2������ɲ������ù������ʵijɷ֣�д���ʵĻ�ѧʽ��������һ��2KMnO4��K2MnO4��MnO2��
�������K2MnO4��MnO2
����ʵ�����ø�����ؼ���Ҳ�ܲ���������С��ͬѧ��16g�ĸ�����ط����Թ��м���һ��ʱ�����ȴ���Ƶ�ʣ�����ʵ�����Ϊ14.4g�����֪���Ϊ����������������Ϊ1.6g��
�⣺�������ص�����Ϊx��
2KMnO4
K2MnO4+MnO2+O2��
2��158 32
x 1.6g
��
=
�����x=15.8g��16g��
�ʾ�������ȷ������ KMnO4��K2MnO4��MnO2����ȷ�ģ�
�ʴ�Ϊ����1�������ǵ�ľ������2������ʯ��ˮû�б仯����3��0.8kg��
��4���ٲ���һ��KMnO4��K2MnO4��MnO2���������K2MnO4��MnO2
��KMnO4��K2MnO4��MnO2
��1����Ϊ������֧��ȼ�գ��ʿ����ô����ǵ�ľ�����ڼ���ƿ�ڼ���������
��2���������ʯ��ˮû�б仯����˵��������̼�ѱ�����������ȫ���գ�
��3����ÿ��ÿ������Ķ�����̼������Ϊ2.2ǧ�ˣ�����ݻ�ѧ����ʽ���Լ��㣮
�⣺�������Ͽɲ�������������Ϊx��
2Na2O2+2CO2=2Na2CO3+O2
88 32
2.2kg x
��
| 88 |
| 2.2kg |
| 32 |
| x |
���������Ͽɲ�������0.8ǧ�ˣ�
��4���ٸ��ݻ�ѧ����ʽ2KMnO4
| ||
�������K2MnO4��MnO2
����ʵ�����ø�����ؼ���Ҳ�ܲ���������С��ͬѧ��16g�ĸ�����ط����Թ��м���һ��ʱ�����ȴ���Ƶ�ʣ�����ʵ�����Ϊ14.4g�����֪���Ϊ����������������Ϊ1.6g��
�⣺�������ص�����Ϊx��
2KMnO4
| ||
2��158 32
x 1.6g
��
| 2��158 |
| x |
| 32 |
| 1.6g |
�ʾ�������ȷ������ KMnO4��K2MnO4��MnO2����ȷ�ģ�
�ʴ�Ϊ����1�������ǵ�ľ������2������ʯ��ˮû�б仯����3��0.8kg��
��4���ٲ���һ��KMnO4��K2MnO4��MnO2���������K2MnO4��MnO2
��KMnO4��K2MnO4��MnO2
����������Ϊ���ݻ�ѧ����ʽ�����������еĻ������㣬�����ļ��㲽�衢���ɵ���д��ʽ�ǽ���������ʱ��Ҫ����ĵط���

��ϰ��ϵ�д�
 ������ϰ�ο����뵥Ԫ���ϵ�д�
������ϰ�ο����뵥Ԫ���ϵ�д� �����Ծ���ĩ���100��ϵ�д�
�����Ծ���ĩ���100��ϵ�д�
�����Ŀ
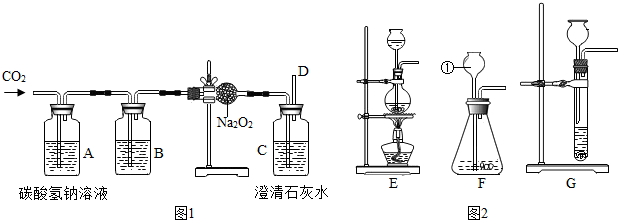
 16g�ĸ�����ط����Թ��м���һ��ʱ�����ȴ���Ƶ�ʣ�����ʵ�����Ϊ14��4 g��
16g�ĸ�����ط����Թ��м���һ��ʱ�����ȴ���Ƶ�ʣ�����ʵ�����Ϊ14��4 g��