��Ŀ����
С����С�֡�С����ͬѧһ��ⶨ˫��ˮ��Һ�����ʵ������������������ϣ�2H2O2
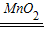 2H2O+O2���������������ܶ�Ϊ1.42g/L��
2H2O+O2���������������ܶ�Ϊ1.42g/L�����ԭ������һ��������˫��ˮ��Ʒ��������̻�ϣ��ⶨ��Ӧ�����������������������H2O2���������̶���˫��ˮ��Һ�����ʵ��������������̷�����
��1��С��������ƣ���ͼ������װ����Ʒ��Һ��20g������ƿ��75g����Ͷ��0.5g�������̣���˫��ˮ��Ӧ��Ϻ�����ƿ�ͷ�Ӧ�������������Ϊ95.1g�������������������g��˫��ˮ��Һ�����ʵ���������Ϊ��
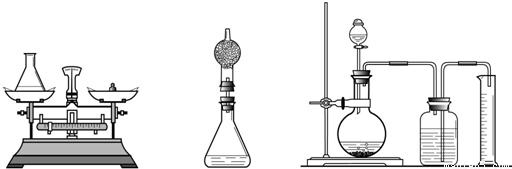
��2��С�ֶ�С�������������ɺ��������ͼ�ķ�����ֻ�����������е���ƿ�ϼ�һ����ܣ�������˵��С�����ɵ������ǣ�
��3��С��ͨ��˼���������ǵķ������ϴ�����С���������ͼ�ķ������Լ��������䣩������˵��С����Ϊ�������ϴ��ԭ�����Ҫ����С����Ҫ��ʵ�����õ���Ͳ��
���𰸡���������1�����������غ㶨�ɣ�������������������������÷�Ӧ�Ļ�ѧ����ʽ����������������������������������������Һ����������������
��2���Ա�����װ���еIJ�ͬ���������ɵ����ɣ�
��3���Ա�װ�÷����������IJ�ͬ��Ȼ���ǰ����װ�ý������ۣ�ͨ���������������ȷ����Ͳ�����̣�
����⣺��1���ų�����������=20g+75g+0.5g-95.1g=0.4g
������0.4g������Ҫ������������Ϊx
2H2O2 2H2O+O2��
2H2O+O2��
68 32
x 0.4g
68��32=x��0.4g ��֮�� x=0.85g
˫��ˮ��Һ�����ʵ���������= =4.25%
=4.25%
�ʴ�Ϊ��0.4g��4.25%��
��2��С��ֻ��С���ķ����е���ƿ�ϼ�һ����ܣ�˵��С����Ϊˮ�ֵ������Բⶨ��Ӱ�죻
�ʴ�Ϊ�����������ˮ������
��3��С������Ƶ�װ�������òⶨ�ų��������������ٲ�ų��������������ˣ�С��Ӧ����Ϊ������������ʱ������������С������ƽ�ľ�ȷ��Ϊ0.1g�����ϴų���������=0.4g÷1.42g/L×1000��282mL����ѡ500mL��Ͳ��
�ʴ�Ϊ��������ƽ�ľ�ȷ��Ϊ0.1g�����ϴ�500mL��
��������Ͳ�ж��ֹ��һ���������Һ�����������ѡ����Ͳ�Ĺ����ѡ����һ����ȡ����ӽ�������������Ͳ����ѡ�����̹�����С����Ͳ�������ײ����ϴ�����Ӷ�Ӱ��ʵ������
��2���Ա�����װ���еIJ�ͬ���������ɵ����ɣ�
��3���Ա�װ�÷����������IJ�ͬ��Ȼ���ǰ����װ�ý������ۣ�ͨ���������������ȷ����Ͳ�����̣�
����⣺��1���ų�����������=20g+75g+0.5g-95.1g=0.4g
������0.4g������Ҫ������������Ϊx
2H2O2
 2H2O+O2��
2H2O+O2��68 32
x 0.4g
68��32=x��0.4g ��֮�� x=0.85g
˫��ˮ��Һ�����ʵ���������=
 =4.25%
=4.25%�ʴ�Ϊ��0.4g��4.25%��
��2��С��ֻ��С���ķ����е���ƿ�ϼ�һ����ܣ�˵��С����Ϊˮ�ֵ������Բⶨ��Ӱ�죻
�ʴ�Ϊ�����������ˮ������
��3��С������Ƶ�װ�������òⶨ�ų��������������ٲ�ų��������������ˣ�С��Ӧ����Ϊ������������ʱ������������С������ƽ�ľ�ȷ��Ϊ0.1g�����ϴų���������=0.4g÷1.42g/L×1000��282mL����ѡ500mL��Ͳ��
�ʴ�Ϊ��������ƽ�ľ�ȷ��Ϊ0.1g�����ϴ�500mL��
��������Ͳ�ж��ֹ��һ���������Һ�����������ѡ����Ͳ�Ĺ����ѡ����һ����ȡ����ӽ�������������Ͳ����ѡ�����̹�����С����Ͳ�������ײ����ϴ�����Ӷ�Ӱ��ʵ������

��ϰ��ϵ�д�
 99��1������ĩ��ѵ��ϵ�д�
99��1������ĩ��ѵ��ϵ�д� ��ǿ��У��ĩ���100��ϵ�д�
��ǿ��У��ĩ���100��ϵ�д� �óɼ�1��1��ĩ���100��ϵ�д�
�óɼ�1��1��ĩ���100��ϵ�д� ��״Ԫ���źþ�ϵ�д�
��״Ԫ���źþ�ϵ�д�
�����Ŀ
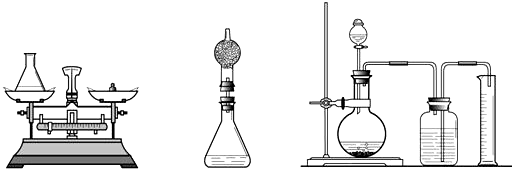

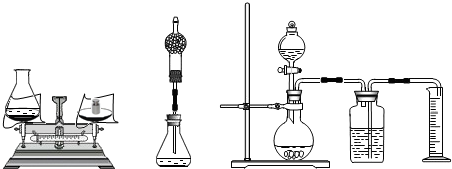
 2H2O+O2���������������ܶ�Ϊ1.42g/L��
2H2O+O2���������������ܶ�Ϊ1.42g/L��
