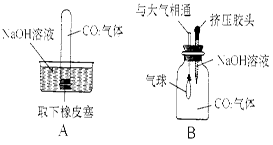��Ŀ����
(4��)Χ��һƿ�������ƹ����Ƿ���ʵ����⣬ͬѧ��չ��̽������������ϣ��������Ʊ�������̼���ƣ�̼������Һ��PH����7��
��1����ͬѧ�÷�̪��Һ�������������Ƿ���ʡ�����Ϊ���ܷ�ɹ� �������ǣ�
��2����ͬѧȡ�����������Թ��У��μ�ij���Լ����д������ݲ������ɴ�֤�����������Ѿ����ʡ�����Ϊ�����Լ�Ϊ�� ��
��3����ͬѧ��֤�����ʵĹ������д����������ƣ���������������̽��������
��1����ͬѧ�÷�̪��Һ�������������Ƿ���ʡ�����Ϊ���ܷ�ɹ� �������ǣ�
��2����ͬѧȡ�����������Թ��У��μ�ij���Լ����д������ݲ������ɴ�֤�����������Ѿ����ʡ�����Ϊ�����Լ�Ϊ�� ��
��3����ͬѧ��֤�����ʵĹ������д����������ƣ���������������̽��������
| ̽��Ŀ�� | ̽������ | ̽������ |
| Ŀ�ģ� | ȡ������������ˮ�������еμ������� ��Һ�� | ������ɫ���� |
| ֤�������д����������� | ������������Һ�еη�̪��Һ | ��̪��Һ��� |
��1�����ܳɹ�����Ϊ����������Һ��ʹ��̪��Һ��졣
��2��ϡ����
��3���Ȼ���
��2��ϡ����
��3���Ȼ���
��ͬѧ��NaOH�ı���ԭ���������˿����е�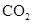 ��������
�������� ����̼������Һ�ʼ��ԣ�����������ҺҲ�ʼ��ԣ���̪�������Һ�����ɫ��
����̼������Һ�ʼ��ԣ�����������ҺҲ�ʼ��ԣ���̪�������Һ�����ɫ��
��ͬѧ��Ҫ֤�����ʾ���֤���� ���ɣ���������������ʺ��д������ݲ�����������ѧ֪ʶ̼�������ᷴӦ�ų�����������̼���壬���Կ���Ϊϡ�����ϡ���ᣮ
���ɣ���������������ʺ��д������ݲ�����������ѧ֪ʶ̼�������ᷴӦ�ų�����������̼���壬���Կ���Ϊϡ�����ϡ���ᣮ
��ͬѧ����������������Һ��̼������Һ���ʼ��ԣ������֤���������ƵĴ��ڣ����ȵ��Ȱ�̼���Ƴ�ȥ����������Ȼ�����Һ��ȥ̼���ƣ����÷�̪��Һ֤���������ƵĴ���
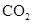 ��������
�������� ����̼������Һ�ʼ��ԣ�����������ҺҲ�ʼ��ԣ���̪�������Һ�����ɫ��
����̼������Һ�ʼ��ԣ�����������ҺҲ�ʼ��ԣ���̪�������Һ�����ɫ����ͬѧ��Ҫ֤�����ʾ���֤����
 ���ɣ���������������ʺ��д������ݲ�����������ѧ֪ʶ̼�������ᷴӦ�ų�����������̼���壬���Կ���Ϊϡ�����ϡ���ᣮ
���ɣ���������������ʺ��д������ݲ�����������ѧ֪ʶ̼�������ᷴӦ�ų�����������̼���壬���Կ���Ϊϡ�����ϡ���ᣮ��ͬѧ����������������Һ��̼������Һ���ʼ��ԣ������֤���������ƵĴ��ڣ����ȵ��Ȱ�̼���Ƴ�ȥ����������Ȼ�����Һ��ȥ̼���ƣ����÷�̪��Һ֤���������ƵĴ���

��ϰ��ϵ�д�
�����Ŀ
 ��1��ʵ��A֤������ȼ�ղ����Ļ��棬�����¶��� ����ߡ��͡��� ��
��1��ʵ��A֤������ȼ�ղ����Ļ��棬�����¶��� ����ߡ��͡��� ��