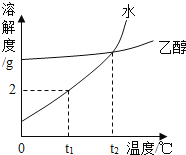
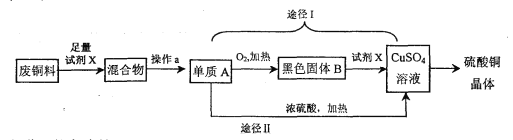
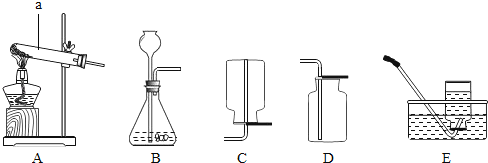
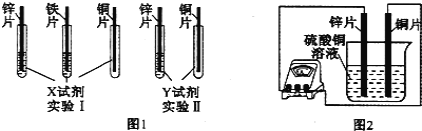
��Ŀ����
����Ŀ���ߴ������������ڵ��ӹ�ҵ��Ҫ��������ԭ�ϣ����������Һ�Ʊ��ߴ������ƵĹ����������£�

��Ӧ���з�����Ӧ�Ļ�ѧ����ʽΪ��CO3+2NH3H2O+Ca��NO3��2�T2NH4NO3+CaCO3��+H2O
��ش��������⣺
��1�����˲����в������������� ��
��2�����˺����ó���������ܺ��еĿ����������� ��д��һ�ּ�������
��3���ù��������п�ѭ��ʹ�õ������� ��
��4����ȴ���ڸ���Ļ����н��У������Ƶõĸߴ��������л���е������� ��
��5�����������еĸ�����NH4NO3���������ʣ����ʵ���Ҫ������ ��
A���ٽ�ֲ��������ҶɫŨ��
B����ǿ����Ŀ�������������
C����ǿ����Ŀ����溦�Ϳ�����������
���𰸡���1������ ��2�������
��3��������̼ ��4���������� ��5��A
��������
�����������1�����˲����в����������������������������
��2�����˺����ó���������ܺ��еĿ���������������ơ�����淋ȣ��������ƣ�
��3����Ϊ̼������ȷֽ������ɶ�����̼�����Ըù��������п�ѭ��ʹ�õ������Ƕ�����̼�����������̼��
��4����ȴ���ڸ���Ļ����н��У������Ƶõĸߴ��������ܺ�ˮ��Ӧ�����������ƣ�����������ƣ�
��5�����������еĸ�����NH4NO3���������ʣ����ʵ���Ҫ�����Ǵٽ�ֲ��������ҶɫŨ�̣����A��