��Ŀ����
��ͼΪij��ѧ��Ӧ����ģ��ͼ������ģ��ͼ�ش�

���С� ����ʾ��ԭ�ӣ���
����ʾ��ԭ�ӣ��� ����ʾ̼ԭ�ӣ���
����ʾ̼ԭ�ӣ��� ����ʾ��ԭ�ӣ�
����ʾ��ԭ�ӣ�
�����ʾ��ͼ�ش��������⣺
��1�������仯�е������������������
��2����H2O2���Ԫ����ͬ����
��3��д����ѧ��Ӧ�ķ���ʽ
��4��������Ӧ����

����
 ����ʾ��ԭ�ӣ���
����ʾ��ԭ�ӣ��� ����ʾ̼ԭ�ӣ���
����ʾ̼ԭ�ӣ��� ����ʾ��ԭ�ӣ�
����ʾ��ԭ�ӣ������ʾ��ͼ�ش��������⣺
��1�������仯�е������������������
C��D
C��D
������ţ���2����H2O2���Ԫ����ͬ����
D
D
����3��д����ѧ��Ӧ�ķ���ʽ
C2H6O+3O2
2CO2+3H2O
| ||
C2H6O+3O2
2CO2+3H2O
��
| ||
��4��������Ӧ����
����
����
��Ӧ����ֽ⡱�����ϡ����û��������ֽ⡱��������֮һ�������������ݻ�ѧ��Ӧ����ģ��ͼ��A�Ļ�ѧʽΪC2H6O��B�Ļ�ѧʽΪO2��C�Ļ�ѧʽΪCO2��D�Ļ�ѧʽΪH2O����Ӧ�Ļ�ѧ����ʽΪC2H6O+3O2
2CO2+3H2O��Ȼ����ݷ�Ӧ�ص��жϷ�Ӧ���ͣ�
| ||
����⣺��1��������ģ���⣬�����仯�е������������������CO2��H2O������C��D��
��2��ˮ�������ⶼ�����⡢��Ԫ����ɵģ�����H2O2���Ԫ����ͬ����D��
��3��������ģ��ͼ���÷�Ӧ�Ļ�ѧ����ʽΪ��C2H6O+3O2
2CO2+3H2O��
��4�����ݻ�ѧ��Ӧ����ʽ���÷�Ӧ�����ϻ��ϡ��û����ֽ���ֽⷴӦ��ֻ����������Ӧ�����������Ӧ��
��2��ˮ�������ⶼ�����⡢��Ԫ����ɵģ�����H2O2���Ԫ����ͬ����D��
��3��������ģ��ͼ���÷�Ӧ�Ļ�ѧ����ʽΪ��C2H6O+3O2
| ||
��4�����ݻ�ѧ��Ӧ����ʽ���÷�Ӧ�����ϻ��ϡ��û����ֽ���ֽⷴӦ��ֻ����������Ӧ�����������Ӧ��
���������ݱ仯��ʾ��ͼ�����ӹ���ģ��ͼ�����ݷ�����ԭ�ӹ��ɵ��ص㣬��ȷ�жϱ仯�е����ʹ��ɣ��ǽ����Ļ���������

��ϰ��ϵ�д�
 �п������п��Ծ����ϵ�д�
�п������п��Ծ����ϵ�д� ��������״Ԫ��ϵ�д�
��������״Ԫ��ϵ�д� �ƸԿ�����ҵ��ϵ�д�
�ƸԿ�����ҵ��ϵ�д�
�����Ŀ
��������AlN�������͵ĵ��Ӿ�Ե��Ƭ���ϣ����ڴ��ͺͳ���
�ͼ��ɵ�·�У��ڸ����µ�����̼��������������Ӧ�������ɵ������������һ����̼���ô˷����Ƶõĵ������к�����������̼����������ijУ��ѧ��ȤС������������Ӧԭ����ȡ���ⶨ������Ʒ�е�����������������
�������ϣ���1��Al2O3+2NaOH=2NaAlO2+H2O������2��AlN+NaOH+H2O=NaAlO2+NH3�������������ᷴӦ��
���ʵ�飺
ʵ��һ����ȡ��������Ʒ
ʵ������ⶨ���Ƶ�������Ʒ�е���������������
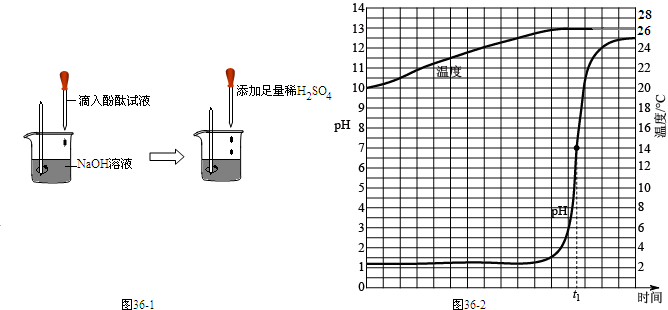
�ٰ�ͼ2��װ��������9.1g��������Ʒ������ƿ�У��ڷ�Һ©���м���һ������ŨNaOH��Һ
�ڳƵ�ʢ��ϡ������ձ���©����������Ϊ200g
�۴�ֹˮ��C���ӷ�Һ©������ƿ�е���ŨNaOH��Һ��ֱ�����ٲ�������Ϊֹ
�ܴ�ֹˮ��B���ӵ���A����������һ�����Ŀ���
���ٴγ���ʢ��ϡ������ձ���©����������Ϊ203.4g
���ظ�ʵ��ܢݲ����Ƶ�ʢ��ϡ������ձ���©������������Ϊ203.4g
ʵ�����ۣ���1������ټ�����Ʒǰ��Ӧ��� ��
��2��װ�м�ʯ�ҵĸ���������� ��װ����ʹ��©����Ŀ���� ��
��3��ͼ2�ձ���ϡ����ܻ���Ũ����������� ��ͼ2�ձ���ϡ����ܻ���Ũ����������� ��ͼ2�ձ���ϡ�����ܷ�ϡ���� ��
��4������ܵ�Ŀ���� ��
ʵ����ۣ����Ƶ�������Ʒ�е���������������Ϊ %��д������̣�4�֣���
�¹ʴ�����ʵ���У�С����С�Ľ�ʢ��Լ50mL��ŨNaOH��Һ���������ϣ���ʱ��Ӧ�ò�ȡ�Ĵ�ʩ�ǣ� ��
��չ̽������1��ʵ���ʣ���ŨNaOH��Һ ���ܻ��ܣ��Ż�ԭ�Լ�ƿ��д��NaOH��Һ¶���ڿ����з����Ļ�ѧ��Ӧ����ʽ ��
��2�����Ž�ԼҩƷ��ԭ��ͬʱ��Ҫȷ��ʵ��˳����óɹ�����ʵ���������Ӧ��40%ŨNaOH��Һ g��д������̣�4�֣���
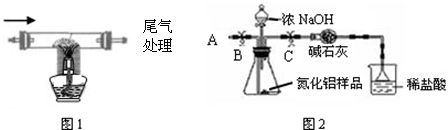
�ͼ��ɵ�·�У��ڸ����µ�����̼��������������Ӧ�������ɵ������������һ����̼���ô˷����Ƶõĵ������к�����������̼����������ijУ��ѧ��ȤС������������Ӧԭ����ȡ���ⶨ������Ʒ�е�����������������
�������ϣ���1��Al2O3+2NaOH=2NaAlO2+H2O������2��AlN+NaOH+H2O=NaAlO2+NH3�������������ᷴӦ��
���ʵ�飺
ʵ��һ����ȡ��������Ʒ
| ʵ�鲽�� | ��ػ�ѧ��Ӧ����ʽ | ||||
| 1����ȥ�����з۳���������̼��������ٳ�ȥ������ | ��ȥ�����Ļ�ѧ��Ӧ����ʽΪ 2Cu+O2
| ||||
| 2���������Ƶõĵ���ͨ��װ��̼������������Ӳ�ʲ������и�����ȡ��������Ʒ����ͼ1���� | װ���з�����ѧ��Ӧ�Ļ�ѧ����ʽΪ N2+3C+Al2O3
|
�ٰ�ͼ2��װ��������9.1g��������Ʒ������ƿ�У��ڷ�Һ©���м���һ������ŨNaOH��Һ
�ڳƵ�ʢ��ϡ������ձ���©����������Ϊ200g
�۴�ֹˮ��C���ӷ�Һ©������ƿ�е���ŨNaOH��Һ��ֱ�����ٲ�������Ϊֹ
�ܴ�ֹˮ��B���ӵ���A����������һ�����Ŀ���
���ٴγ���ʢ��ϡ������ձ���©����������Ϊ203.4g
���ظ�ʵ��ܢݲ����Ƶ�ʢ��ϡ������ձ���©������������Ϊ203.4g
ʵ�����ۣ���1������ټ�����Ʒǰ��Ӧ���
��2��װ�м�ʯ�ҵĸ����������
��3��ͼ2�ձ���ϡ����ܻ���Ũ�����������
��4������ܵ�Ŀ����
ʵ����ۣ����Ƶ�������Ʒ�е���������������Ϊ
�¹ʴ�����ʵ���У�С����С�Ľ�ʢ��Լ50mL��ŨNaOH��Һ���������ϣ���ʱ��Ӧ�ò�ȡ�Ĵ�ʩ�ǣ�
��չ̽������1��ʵ���ʣ���ŨNaOH��Һ
��2�����Ž�ԼҩƷ��ԭ��ͬʱ��Ҫȷ��ʵ��˳����óɹ�����ʵ���������Ӧ��40%ŨNaOH��Һ

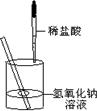 37����ͼ��ʾ��ijͬѧ�ڽ�������кͷ�Ӧ��ʵ��ʱ�����ձ��е�����������Һ�μ�ϡ����һ������������˵μ�ָʾ����Ϊ��ȷ�����������������Ƿ���ȫ��Ӧ����ͬѧ���ձ���ȡ������Ӧ�����Һ��һ֧�Թ��У������Թ��еμӼ�����ɫ��̪��Һ�����۲쵽��̪��Һ����ɫ���������ó�������������ǡ����ȫ�к͡��Ľ��ۣ�
37����ͼ��ʾ��ijͬѧ�ڽ�������кͷ�Ӧ��ʵ��ʱ�����ձ��е�����������Һ�μ�ϡ����һ������������˵μ�ָʾ����Ϊ��ȷ�����������������Ƿ���ȫ��Ӧ����ͬѧ���ձ���ȡ������Ӧ�����Һ��һ֧�Թ��У������Թ��еμӼ�����ɫ��̪��Һ�����۲쵽��̪��Һ����ɫ���������ó�������������ǡ����ȫ�к͡��Ľ��ۣ�
 ij�о�С����ʵ������ȡ������̼���о��з��֣�ͨ����ѡ�ô���ʯ��ϡ���ᷴӦ��ȡ������̼����ԭ���Ƿ�Ӧ������������︲���ڹ���ı��棬�谭�˴���ʯ��ϡ����ĽӴ���Ӧ��������ֹͣ��
ij�о�С����ʵ������ȡ������̼���о��з��֣�ͨ����ѡ�ô���ʯ��ϡ���ᷴӦ��ȡ������̼����ԭ���Ƿ�Ӧ������������︲���ڹ���ı��棬�谭�˴���ʯ��ϡ����ĽӴ���Ӧ��������ֹͣ��