��Ŀ����
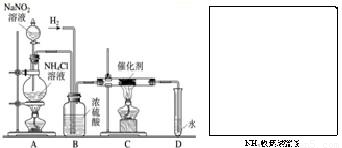
ʵ���ҳ����������ƣ�NaNO2�����Ȼ����Һ��NH4Cl����Ӧ��ȡN2��N2�Ļ�ѧ����ʮ���ȶ�������һ������������H2���ֻ�������NH3����ͼΪ��ȡ����NH3������NH3��װ�ã���ȡH2��װ������ȥ������1��NaNO2��һ�ֹ�ҵ���Σ���______����ܡ����ܡ�������ʳ��ʹ�ã�Aװ��������NaNO2��Һ������������______��
��2��Cװ�õ�Ӳ���Թ��з�����Ӧ�Ļ�ѧ����ʽΪ______��2NH3
���𰸡���������1�������������ƵĶ��Խ��з������˽ⳣ�����������ƺ���;��
��2�����������֪��������������C��������Ӧ���ɰ�����д����Ӧ����ʽ����Ӧ��N2��H2����������Ⱦ��Ƿ������ж��ߵ������ȣ�����ʵ�ʵķ�ӳ�������D�е��ܿ����������ɣ����ݰ��������ʽ�����֤��
��3�����ݰ����ռ�����ȷ���ռ�װ�ã�
��4������Ũ�������ˮ�Է�����
��5�����ٸ��ݷ�Ӧ�������ȼ�ת���ʼ��㣻
�ڸ���п��ϡ���ᷴӦ�������������ķ�Ӧ���ɰ������������п��������
����⣺��1���������������ж��������ܵ���ʳ��ʹ�ã�Aװ��������NaNO2��Һ�������Ƿ�Һ©����
��2��������������C��������Ӧ���ɰ�������Ӧ�ķ���ʽ�ǣ�N2+3H2 2NH3����ѧ����ʽ�����ʵ������������ʵ���������ȣ�Ϊ��14��3�������ǡ����ֻ��ϡ����Ի���δ��Ӧ��H2��N2�ݳ������ԣ�D�е��ܿڻ��ݳ������ݣ����ڰ�����ˮ��Һ�Լ��ԣ����ԣ���֤��NH3���ɵ�ʵ���ǣ���D�е�����ɫ��̪��Һ����̪��죻
2NH3����ѧ����ʽ�����ʵ������������ʵ���������ȣ�Ϊ��14��3�������ǡ����ֻ��ϡ����Ի���δ��Ӧ��H2��N2�ݳ������ԣ�D�е��ܿڻ��ݳ������ݣ����ڰ�����ˮ��Һ�Լ��ԣ����ԣ���֤��NH3���ɵ�ʵ���ǣ���D�е�����ɫ��̪��Һ����̪��죻
��3���ɰ�����������ˮ���ܶȱȿ���С��Ӧ�������ſ������ռ���װ����ͼ��
��4������Ũ���������ˮ�ԣ����������ﰱ����������
��5������N2+3H2 2NH3�ĸ����ʵ�������Ϊ28��6��34=14��3��17��N2��ƽ������Ϊ1.4m g/s��H2������Ϊ0.3m g/s��N2��H2�ڷ�Ӧ��ֻ��ת��40%��֪������NH3��ƽ������Ϊ��1.7m g/s×40%=0.68m g/s��
2NH3�ĸ����ʵ�������Ϊ28��6��34=14��3��17��N2��ƽ������Ϊ1.4m g/s��H2������Ϊ0.3m g/s��N2��H2�ڷ�Ӧ��ֻ��ת��40%��֪������NH3��ƽ������Ϊ��1.7m g/s×40%=0.68m g/s��
����������Ҫ����Zn������ΪX
����ȡ������Ӧ��Zn+H2SO4=ZnSO4+H2����N2+3H2 2NH3���ó�п�백���Ĺ�ϵʽΪ��
2NH3���ó�п�백���Ĺ�ϵʽΪ��
3Zn��2NH3
195 34
X 1.36mg
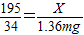 ��ã�X=7.8mg
��ã�X=7.8mg
�ʴ�Ϊ����1�����ܣ���Һ©������2��N2+3H2 2NH3��14��3���ᣬ��D�е�����ɫ��̪��Һ����̪��죻��3������ͼ����4�����ﵪ����������������5����0.68m g/s�� ��7.8mg��
2NH3��14��3���ᣬ��D�е�����ɫ��̪��Һ����̪��죻��3������ͼ����4�����ﵪ����������������5����0.68m g/s�� ��7.8mg��
���������⿼�鰱����ʵ�����Ʒ��������ļ��顢Ũ��������ʵ����ݣ�������ѧ֪ʶ��֪ʶ����Ǩ�Ƽ��ɽ�����⣮
��2�����������֪��������������C��������Ӧ���ɰ�����д����Ӧ����ʽ����Ӧ��N2��H2����������Ⱦ��Ƿ������ж��ߵ������ȣ�����ʵ�ʵķ�ӳ�������D�е��ܿ����������ɣ����ݰ��������ʽ�����֤��
��3�����ݰ����ռ�����ȷ���ռ�װ�ã�
��4������Ũ�������ˮ�Է�����
��5�����ٸ��ݷ�Ӧ�������ȼ�ת���ʼ��㣻
�ڸ���п��ϡ���ᷴӦ�������������ķ�Ӧ���ɰ������������п��������
����⣺��1���������������ж��������ܵ���ʳ��ʹ�ã�Aװ��������NaNO2��Һ�������Ƿ�Һ©����
��2��������������C��������Ӧ���ɰ�������Ӧ�ķ���ʽ�ǣ�N2+3H2
 2NH3����ѧ����ʽ�����ʵ������������ʵ���������ȣ�Ϊ��14��3�������ǡ����ֻ��ϡ����Ի���δ��Ӧ��H2��N2�ݳ������ԣ�D�е��ܿڻ��ݳ������ݣ����ڰ�����ˮ��Һ�Լ��ԣ����ԣ���֤��NH3���ɵ�ʵ���ǣ���D�е�����ɫ��̪��Һ����̪��죻
2NH3����ѧ����ʽ�����ʵ������������ʵ���������ȣ�Ϊ��14��3�������ǡ����ֻ��ϡ����Ի���δ��Ӧ��H2��N2�ݳ������ԣ�D�е��ܿڻ��ݳ������ݣ����ڰ�����ˮ��Һ�Լ��ԣ����ԣ���֤��NH3���ɵ�ʵ���ǣ���D�е�����ɫ��̪��Һ����̪��죻��3���ɰ�����������ˮ���ܶȱȿ���С��Ӧ�������ſ������ռ���װ����ͼ��

��4������Ũ���������ˮ�ԣ����������ﰱ����������
��5������N2+3H2
 2NH3�ĸ����ʵ�������Ϊ28��6��34=14��3��17��N2��ƽ������Ϊ1.4m g/s��H2������Ϊ0.3m g/s��N2��H2�ڷ�Ӧ��ֻ��ת��40%��֪������NH3��ƽ������Ϊ��1.7m g/s×40%=0.68m g/s��
2NH3�ĸ����ʵ�������Ϊ28��6��34=14��3��17��N2��ƽ������Ϊ1.4m g/s��H2������Ϊ0.3m g/s��N2��H2�ڷ�Ӧ��ֻ��ת��40%��֪������NH3��ƽ������Ϊ��1.7m g/s×40%=0.68m g/s������������Ҫ����Zn������ΪX
����ȡ������Ӧ��Zn+H2SO4=ZnSO4+H2����N2+3H2
 2NH3���ó�п�백���Ĺ�ϵʽΪ��
2NH3���ó�п�백���Ĺ�ϵʽΪ��3Zn��2NH3
195 34
X 1.36mg
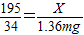 ��ã�X=7.8mg
��ã�X=7.8mg�ʴ�Ϊ����1�����ܣ���Һ©������2��N2+3H2
 2NH3��14��3���ᣬ��D�е�����ɫ��̪��Һ����̪��죻��3������ͼ����4�����ﵪ����������������5����0.68m g/s�� ��7.8mg��
2NH3��14��3���ᣬ��D�е�����ɫ��̪��Һ����̪��죻��3������ͼ����4�����ﵪ����������������5����0.68m g/s�� ��7.8mg�����������⿼�鰱����ʵ�����Ʒ��������ļ��顢Ũ��������ʵ����ݣ�������ѧ֪ʶ��֪ʶ����Ǩ�Ƽ��ɽ�����⣮

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��������Ҫ�ɷ��ǵ��������������е������Լռ���������4/5��
��1�������������������������������������ɫ����ô�����ͻ�����ɫ������������ɫ�ģ������Ʋ���������ɫ�ģ�������������飬�Ե����������ʽ����Ʋ⣬��д�±���
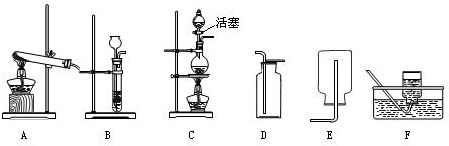
��2��ʵ���ҿ����������ƣ�NaNO2��������Ȼ�泥�NH4Cl����Һ��ͼ�������ȡ���������ṩ����ʵ��װ�ã���ʵ������ȡ����Ӧ���õķ���װ���� ���ռ�װ���� ��������ĸ��
��3����ҵ�ϳ��õ����������ڸ��¡���ѹ�����������ºϳɰ�������ѧ��Ӧ����ʽΪ ��
N2+3H2
2NH3��������ȡ����1.7�֣���������Ҫ��0��ͱ�����ѹ�µĵ���������������֪O��ͱ�����ѹ�µ������ܶ�Ϊ1.25��/�������赪����ȫת��Ϊ������
��1�������������������������������������ɫ����ô�����ͻ�����ɫ������������ɫ�ģ������Ʋ���������ɫ�ģ�������������飬�Ե����������ʽ����Ʋ⣬��д�±���
| �� ɫ | �� ζ | ˮ �� �� | |
| �� �� |

��3����ҵ�ϳ��õ����������ڸ��¡���ѹ�����������ºϳɰ�������ѧ��Ӧ����ʽΪ
N2+3H2
| ||
| ���¸�ѹ |
��2004?���ˣ���������Ҫ�ɷ��ǵ��������������е������Լռ���������4/5��
��1�������������������������������������ɫ����ô�����ͻ�����ɫ������������ɫ�ģ������Ʋ���������ɫ�ģ�������������飬�Ե����������ʽ����Ʋ⣬��д�±���
��2��ʵ���ҿ����������ƣ�NaNO2��������Ȼ�泥�NH4Cl����Һ��ͼ�������ȡ���������ṩ����ʵ��װ�ã���ʵ������ȡ����Ӧ���õķ���װ���ǣ��ռ�װ���ǣ�������ĸ��
��3����ҵ�ϳ��õ����������ڸ��¡���ѹ�����������ºϳɰ�������ѧ��Ӧ����ʽΪ��
N2+3H2 2NH3��������ȡ����1.7�֣���������Ҫ��0��ͱ�����ѹ�µĵ���������������֪O��ͱ�����ѹ�µ������ܶ�Ϊ1.25��/�������赪����ȫת��Ϊ������
2NH3��������ȡ����1.7�֣���������Ҫ��0��ͱ�����ѹ�µĵ���������������֪O��ͱ�����ѹ�µ������ܶ�Ϊ1.25��/�������赪����ȫת��Ϊ������
��1�������������������������������������ɫ����ô�����ͻ�����ɫ������������ɫ�ģ������Ʋ���������ɫ�ģ�������������飬�Ե����������ʽ����Ʋ⣬��д�±���
| �� ɫ | �� ζ | ˮ �� �� | |
| �� �� |

��3����ҵ�ϳ��õ����������ڸ��¡���ѹ�����������ºϳɰ�������ѧ��Ӧ����ʽΪ��
N2+3H2
 2NH3��������ȡ����1.7�֣���������Ҫ��0��ͱ�����ѹ�µĵ���������������֪O��ͱ�����ѹ�µ������ܶ�Ϊ1.25��/�������赪����ȫת��Ϊ������
2NH3��������ȡ����1.7�֣���������Ҫ��0��ͱ�����ѹ�µĵ���������������֪O��ͱ�����ѹ�µ������ܶ�Ϊ1.25��/�������赪����ȫת��Ϊ������  ʵ���ҳ����������ƣ�NaNO2�����Ȼ����Һ��NH4Cl����Ӧ��ȡN2��N2�Ļ�ѧ����ʮ���ȶ�������һ������������H2���ֻ�������NH3����ͼΪ��ȡ����NH3������NH3��װ�ã���ȡH2��װ������ȥ����
ʵ���ҳ����������ƣ�NaNO2�����Ȼ����Һ��NH4Cl����Ӧ��ȡN2��N2�Ļ�ѧ����ʮ���ȶ�������һ������������H2���ֻ�������NH3����ͼΪ��ȡ����NH3������NH3��װ�ã���ȡH2��װ������ȥ����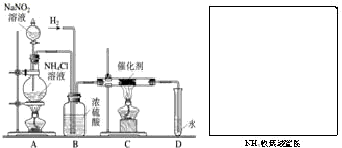 ʵ���ҳ����������ƣ�NaNO2�����Ȼ����Һ��NH4Cl����Ӧ��ȡN2��N2�Ļ�ѧ����ʮ���ȶ�������һ������������H2���ֻ�������NH3����ͼΪ��ȡ����NH3������NH3��װ�ã���ȡH2��װ������ȥ����
ʵ���ҳ����������ƣ�NaNO2�����Ȼ����Һ��NH4Cl����Ӧ��ȡN2��N2�Ļ�ѧ����ʮ���ȶ�������һ������������H2���ֻ�������NH3����ͼΪ��ȡ����NH3������NH3��װ�ã���ȡH2��װ������ȥ����