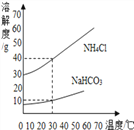
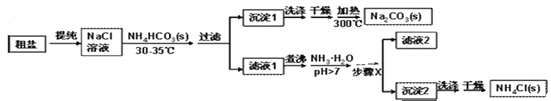
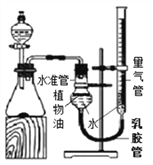
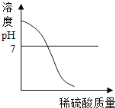
��Ŀ����
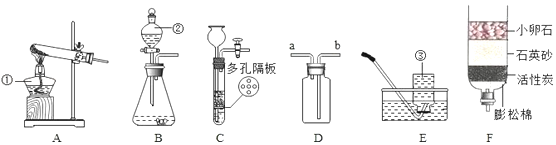
����Ŀ����ͼ��ʵ���ҵIJ���ʵ��װ��ͼ���ش��й����⣺
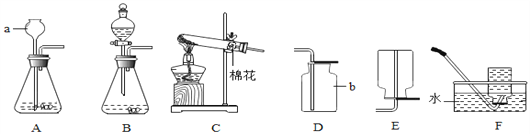
��1��д���������ƣ�a��_______________________��
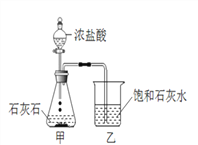
��2����Aװ����ȡ������̼ʱ����©������Һ�����µ�Ŀ����__________________��A�з�����Ӧ�Ļ�ѧ����ʽΪ_____________________________________���ռ�������̼Ӧѡ�õ�װ����________������ţ���
��3����ȡ������̼ʵ�����ʱ��С��ͬѧ��Aװ����ƿ�е���Һ���ײ�û�й��壩�����ձ��У�����Һ�е����ʽ�����̽����
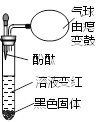
�����⣩��Һ�����ʵijɷ�����Щ��
�����룩С�յIJ��룺��Һ�е��������Ȼ���
С���IJ��룺��Һ�е�������____________________��
��ʵ��֤����С�����Թ�ȡ������Һ���ֱ�����Һ�м��������Լ��е�______������ţ������û�����������֤��С�յIJ�����ȷ��
������������Һ ��ʯ����Һ �۷�̪��Һ �ܴ���ʯ
���𰸡� ����©�� ��ֹ������̼�ӳ���©���ݳ� CaCO3+2HCl=CaCl2+H2O+CO2�� D CaCl2��HCl �ڢ�
����������1��a�dz���©������2����Aװ����ȡ������̼ʱ����©������Һ�����µ�Ŀ���Ƿ�ֹ������̼�ӳ���©���ݳ���A�з�����Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HCl=CaCl2+H2O+CO2����������̼������ˮ����������ˮ���ռ���������̼���ܶȱȿ������ܶȴ��������ſ������ռ�����ѡD��(3)�����Ӧʱϡ����ʣ�࣬��������CaCl2��HCl��������������Һ��ϡ���ᷴӦ�����������������⣻��ʯ����Һ������Һ��죬�������⣻�۷�̪��Һ��������Һ����ɫ�����������⣻�ܴ���ʯ��ϡ���ᷴӦ�����������ɶ�ð���ݣ��������⡣��ѡ�ڢܡ�

����Ŀ����ʡú̿��Դ�ḻ����˹������ú�㼰��Χ�Ҳ��У��Ǿ����к�������ܳƣ���Ҫ�ɷ��Ǽ��顣
(1)����д��������ȫȼ�յĻ�ѧ����ʽ��__________________________________��
(2)ú����˹��ը��������Ҫ������
����˹�����ڱ�ը���ķ�Χ�ڣ���________________________________________��
(3)�±��dz�����������ı�ը���ޣ�����ݴ��жϣ�����������ը��������____��
��ȼ���� | ��ը���ޣ���������� |
H2 | 4.0%~74.2% |
CH4 | 5%~15% |
CO | 12.5%~74.2% |
(4)����ͼ���У���ȼ�պͱ�ը�ص���____��

(5)ij�ִ����Ŀ����ˡ���Ũ����˹���缼��������Ч����˿������ܱߵ����������õ硣���ּȲ�ú�ַ��������������ŵ���___________________________________��