��Ŀ����
2013����4��7�ա�����ֱ���䡷��Ŀ�������ӱ����غ�ɫ����ˮ�¼��ٱ�������70������ӱ�ʡ�����в����Ź�����С��ׯdz�����ˮ���ֺ�ɫ�����ص�����ֻ����Ϊ�����������ĵ���ˮ���������¼���
��1���Ӹй�ָ�����жϴ�ˮ ����ǡ����ǡ����ϸ������ˮ��Ϊ�˳�ȥˮ���е���ɫ����ζ���Լ��� ��
��2�������pH��ֽ������ˮ������ ��
��3����Ҫ�жϴ�ˮ�Dz���Ӳˮ�������� ���ճ������пɲ��� �ķ�����Ӳˮ������
��4���������������ˮ����Ⱦ���� ��
A�����ⶪ���Ͼɵ�ء�B����SO2�Ĺ�ҵ�����ŷ�
C����ҵ��ˮ͵͵�ŷš� D��������ˮֱ���ŷţ�
��1���Ӹй�ָ�����жϴ�ˮ
��2�������pH��ֽ������ˮ������
��3����Ҫ�жϴ�ˮ�Dz���Ӳˮ��������
��4���������������ˮ����Ⱦ����
A�����ⶪ���Ͼɵ�ء�B����SO2�Ĺ�ҵ�����ŷ�
C����ҵ��ˮ͵͵�ŷš� D��������ˮֱ���ŷţ�
���㣺����ˮ�����������뾻������,��Һ�����Ȳⶨ,Ӳˮ����ˮ,ˮ��Դ����Ⱦ�����
ר�⣺������ˮ
��������1���ݵ���ˮ����ɫ�ͻ���̿�������Է�����
��2���ݲⶨ��Һ���ȵķ���������
��3��Ӳˮ�����ˮ��ϲ�������������ˮ��ϲ�����ĭ��Ӳˮ�������ǽ���ˮ�п����Ը�þ������ĺ�����
��4��A�����ݵ���к����ؽ������������ⶪ�������ˮ����Ⱦ���н��
B���ݶ��������ܹ��γ������Ǵ�����Ⱦ�������
C�����ݹ�ҵ��ˮ�к����к����ʣ���ҵ��ˮ�ŷŻ����ˮ����Ⱦ���н��
D������������ˮ��ֱ���ŷ��ܹ����ˮ����Ⱦ���н��
��2���ݲⶨ��Һ���ȵķ���������
��3��Ӳˮ�����ˮ��ϲ�������������ˮ��ϲ�����ĭ��Ӳˮ�������ǽ���ˮ�п����Ը�þ������ĺ�����
��4��A�����ݵ���к����ؽ������������ⶪ�������ˮ����Ⱦ���н��
B���ݶ��������ܹ��γ������Ǵ�����Ⱦ�������
C�����ݹ�ҵ��ˮ�к����к����ʣ���ҵ��ˮ�ŷŻ����ˮ����Ⱦ���н��
D������������ˮ��ֱ���ŷ��ܹ����ˮ����Ⱦ���н��
����⣺��1���˵���ˮΪ��ɫ���ʴ�ˮ���Ǻϸ������ˮ������̿���������ԣ���������ˮ�е�ɫ�غ���ζ��
��2����pH��ֽ������ˮ�����ȵķ����ǣ��ò�����պȡˮ����pH��ֽ�ϣ�����ֽ���ֵ���ɫ�����ɫ�����գ�
��3������Ӳˮ����ˮʹ�õ��Ƿ���ˮ������ˮ��Ӳˮ��ϲ�������������ˮ��ϲ�����ĭ��Ӳˮ�������ǽ���ˮ�п����Ը�þ������ĺ����������Ը�þ�����������ײ��������Ը�þ����������п���ʹ�ü�����еķ�����Ӳˮ������
��4��A������к����ؽ������������ⶪ�������ˮ����Ⱦ��
B�����������ܹ��γ����꣬��Ⱦˮ�壻C����ҵ��ˮ�к����к����ʣ���ҵ��ˮ�ŷŻ����ˮ����Ⱦ��
D��������ˮ��ֱ���ŷ��ܹ����ˮ����Ⱦ��
��ѡ��ABCD��
�ʴ�Ϊ����1�����ǡ�����̿��2���ò�����պȡˮ����pH��ֽ�ϣ�����ֽ���ֵ���ɫ�����ɫ�����գ���3������ˮ����С���4��ABCD��ÿ��һ�֣���
��2����pH��ֽ������ˮ�����ȵķ����ǣ��ò�����պȡˮ����pH��ֽ�ϣ�����ֽ���ֵ���ɫ�����ɫ�����գ�
��3������Ӳˮ����ˮʹ�õ��Ƿ���ˮ������ˮ��Ӳˮ��ϲ�������������ˮ��ϲ�����ĭ��Ӳˮ�������ǽ���ˮ�п����Ը�þ������ĺ����������Ը�þ�����������ײ��������Ը�þ����������п���ʹ�ü�����еķ�����Ӳˮ������
��4��A������к����ؽ������������ⶪ�������ˮ����Ⱦ��
B�����������ܹ��γ����꣬��Ⱦˮ�壻C����ҵ��ˮ�к����к����ʣ���ҵ��ˮ�ŷŻ����ˮ����Ⱦ��
D��������ˮ��ֱ���ŷ��ܹ����ˮ����Ⱦ��
��ѡ��ABCD��
�ʴ�Ϊ����1�����ǡ�����̿��2���ò�����պȡˮ����pH��ֽ�ϣ�����ֽ���ֵ���ɫ�����ɫ�����գ���3������ˮ����С���4��ABCD��ÿ��һ�֣���
���������⿼����ˮ�ľ����Լ���ˮ��Ӳˮ�ļ�����ɴ��⣬�����������е�֪ʶ���У�

��ϰ��ϵ�д�
�����Ŀ
���л�ѧ����ʽ�У���ȷ���ǣ�������
| A��CuO+H2O�TCu��OH��2 |
| B��Mg��OH��2+2HCl�TMgCl2+2H2O |
| C��Zn��NO3��2+2KCl�TZnCl2+2KNO3 |
| D��Cu+ZnSO4�TCuSO4+Zn |
���й��ڳ�����һЩ����Ľ��Ͳ���ȷ���ǣ�������
| A���ŵ��������ζ������Ϊ���ӵ����������� |
| B���տ�ˮʱ���DZ���������Ϊ���Ӽ������ |
| C����ѹ��������Ŀ죬����Ϊѹǿ���е����� |
| D���õ��Ρ���������Ŀ����Ϊ�˲����Ԫ�ء���Ԫ�� |
ijʵ��С��������ͼ��ʾ������������һ����������������������Һ��ʵ�飬��ʵ��Ҫ��ͼ�л�ȱ�ٵIJ��������ǣ�������

A�� ©�� |
B�� �Թ� |
C�� ������ |
D�� ����ƿ |
 2014��8�£���»Ὣ���Ͼ����У�
2014��8�£���»Ὣ���Ͼ����У�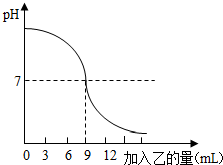 ��֪�ס��ҷֱ���������Һ������������Һ�е�һ�֣���ͼ��ʾ����ʢ��10mLϡ�ļ���Һ�����е���������ɫʯ���Լ����У���������Һʱ����ҺpH�ı仯���ߣ���������ش����⣺
��֪�ס��ҷֱ���������Һ������������Һ�е�һ�֣���ͼ��ʾ����ʢ��10mLϡ�ļ���Һ�����е���������ɫʯ���Լ����У���������Һʱ����ҺpH�ı仯���ߣ���������ش����⣺