��Ŀ����
С����ʵ���ҷ���һƿ�������ƹ���û������ƿ����������������ͬѧһ�����ƿ�������ƹ���չ��̽��������������һ�����̽����
��������⡿��ƿ�������ƹ����Ƿ���ʣ�
���������ϡ�CaCl2��Һ�����ԡ�
����������衿
����������û�б��ʣ��ڸ��������Ʋ��ֱ��ʣ��۸���������??????? ��
�����ʵ�顿ȡ�������ƹ�����Ʒ����ˮ�����ҺA����������ʵ�飺
ʵ�鲽�� | ʵ������ | ���ۼ����� |
��1��ȡ������ҺA���Թ��У������еμ�������ϡ��� | �����ݲ��� | ����????????? ������ |
��2����ȡ������ҺA���Թ��У������еμ��������Ȼ�����Һ�� | ����???? ???? | ��Ӧ�Ļ�ѧ����ʽΪ��
|
��3�������裨2�����õĻ��Һ���ã����ϲ���Һ�еμ���ɫ�ķ�̪��Һ�� | ��Һ�ʺ�ɫ | ����???????? ���� |
����˼�뽻����
���õ��������Ʊ��ʵ�ԭ���ǣ��û�ѧ����ʽ��ʾ��?????????????????? ����ˣ���������Ӧ???????? ���档
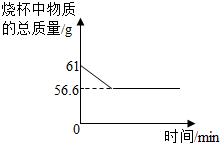
��̽������չ��Ϊ��һ��̽���������Ƶı��ʳ̶ȣ�С����ȡ18.6g�������ƹ�����Ʒ�����ձ��У���������μ���ϡ���ᣬ�����ٲ�������Ϊֹ��������ϡ����100g����Ӧ��Ƶ��ձ�����Һ������Ϊ114.2g���Լ��㣺
��1������Ʒ��̼���Ƶ�����Ϊ??????????????? ��
��2�����������Ƶı��ʳ̶ȣ������ʵ���������ռ����ǰ���������Ƶ�����������Ϊ?????????????? ��
����������衿��ȫ������
�����ʵ�顿
ʵ�鲽�� | ʵ������ | ���ۼ����� |
��1�� |
| �� |
��2�� | ��ɫ���� | Na2CO3+ CaCl2=CaCO3��+2NaCl |
��3�� |
| �� |
����˼�뽻����CO2+ 2NaOH = Na2CO3 + H2O�� �ܷ�
��̽������չ����1��10.6?? ��2��50%?????????????
��������
�������������������衿��������������������û�б��ʣ��ڸ��������Ʋ��ֱ��ʣ��۸���������ȫ������
�����ʵ�顿����Ϊ̼���������ᷴӦ���ɶ�����̼���������ݣ�Na2CO3+2HCl=2NaCl+H2O+CO2����������������Ȼ��Ҳ���ᷴӦ����û���������ɣ�NaOH+HCl=NaCl+H2O�����Ը������������ݲ�����˵����̼���ƴ��ڣ��ʲ���ٲ��������ڲ��裨2���еμ��������Ȼ�����Һ����Һ�е�̼���ƻ����Ȼ��Ʒ�Ӧ��CaCl2+Na2CO3=CaCO3��+2NaCl�����������˰�ɫ�������������ӵ��Ȼ�����Һ��������������Һ�е�̼����ȫ����Ӧ���ˣ����ڲ��裨3���н����裨2�����õĻ��Һ���ã����ϲ���Һ�еμ���ɫ�ķ�̪��Һ�������Һ�ʺ�ɫ��˵����Һ��Ȼ�ʼ��ԣ�Ҳ����˵��Һ����NaOH�����Բ���ڳ�������Һ���ֱ���
����˼�뽻���������õ��������������տ����еĶ�����̼�����ʣ���ѧ����ʽΪ��CO2+ 2NaOH = Na2CO3 + H2O����ˣ���������Ӧ�ܷⱣ��
��̽������չ�������������غ㶨�����μ���ѧ��Ӧ�ĸ����ʵ������ܺͣ����ڷ�Ӧ�������ĸ����ʵ������ܺͣ����Կ���ֱ���ж����ɵĶ�����̼��������=18.6g+100g-114.2g=4.4g���ٸ��ݻ�ѧ��Ӧ����ʽ��Na2CO3+2HCl=2NaCl+H2O+CO2���ж�����̼��̼���Ƶ�������ϵ������̼���Ƶ�������Ȼ���ٸ��ݻ�ѧ��Ӧ����ʽ��CO2+ 2NaOH = Na2CO3 + H2O��̼������NaOH��������ϵ������ʵ�NaOH��������һ����������������Ƶı��ʳ̶�
�⣺��̼���Ƶ�����Ϊx�����ʵ�NaOH����Ϊy
Na2CO3+2HCl=2NaCl+H2O+CO2��
106????????????????????? 44
x?????????????????????? 4.4g
��1��106��44=x��4.4g???? ��x=10.6g??
��ԭ������δ���ʵ�NaOH����=18.6g-10.6g=8g
CO2+ 2NaOH = Na2CO3 + H2O
?????? 80????? 106
?????? y???? 10.6g
��2��80��106=y��10.6g????? ��y=8g
�����������Ƶı��ʳ̶�=8g��(8g+8g)��100%=50%
���㣺�������Ƶı��ʵ�̽��,̼���ƵĻ�ѧ����,���ݻ�ѧ����ʽ���еļ���

С����ʵ���ҷ���һƿ�������ƹ���û������ƿ����������������ͬѧһ�����ƿ�������ƹ���չ��̽��������������һ�����̽����
��������⡿��ƿ�������ƹ����Ƿ���ʣ�
���������ϡ�CaCl2��Һ�����ԡ�
����������衿
����������û�б��ʣ��ڸ��������Ʋ��ֱ��ʣ��۸��������� ��
�����ʵ�顿ȡ���� ���ƹ�����Ʒ����ˮ�����ҺA����������ʵ�飺
���ƹ�����Ʒ����ˮ�����ҺA����������ʵ�飺
| ʵ�鲽�� | ʵ������ | ���ۼ����� |
| ��1��ȡ������ҺA���Թ��У������еμ�������ϡ��� | �����ݲ��� | ���� ������ |
| ��2����ȡ������ҺA���Թ��У������еμ��������Ȼ�����Һ�� | ���� | ��Ӧ�Ļ�ѧ����ʽΪ�� |
| ��3�������裨2�����õĻ��Һ���ã����ϲ���Һ�еμ���ɫ�ķ�̪��Һ�� | ��Һ�ʺ�ɫ | ���� ���� |
����˼�뽻����
���õ��������Ʊ��ʵ�ԭ���ǣ��û�ѧ����ʽ��ʾ�� ����ˣ���������Ӧ ���档
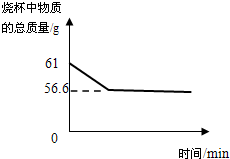
��̽������չ��Ϊ��һ��̽���������Ƶı��ʳ̶ȣ�С����ȡ18.6g�������ƹ�����Ʒ�����ձ��У���������μ���ϡ���ᣬ�����ٲ�������Ϊֹ��������ϡ����100g����Ӧ��Ƶ��ձ�����Һ������Ϊ114.2g���Լ��㣺
��1������Ʒ��̼���Ƶ�����Ϊ ��
��2�����������Ƶı��ʳ̶ȣ������ʵ���������ռ����ǰ���������Ƶ�����������Ϊ
��
 ��2012?������ģ��С����ʵ���ҷ���һƿʢ����ɫ��Һ���Լ�ƿ�����ǩ���𣬴Ӳ��ಿ��ֻ�ܿ���������������Ϊ10%��������ʲô���������ϣ���ʦ����������ƿ��Һ�������������ơ��Ȼ��ơ��������ƻ���̼�����е�һ�֣�
��2012?������ģ��С����ʵ���ҷ���һƿʢ����ɫ��Һ���Լ�ƿ�����ǩ���𣬴Ӳ��ಿ��ֻ�ܿ���������������Ϊ10%��������ʲô���������ϣ���ʦ����������ƿ��Һ�������������ơ��Ȼ��ơ��������ƻ���̼�����е�һ�֣�
 С����ʵ���ҷ���һƿʢ����ɫ��Һ���Լ�ƿ�����ǩ���𣬴Ӳ��ಿ��ֻ�ܿ���������������Ϊ10%��������ʲô���������ϣ���ʦ����������ƿ��Һ�������������ơ��Ȼ��ơ��������ƻ���̼�����е�һ�֣�
С����ʵ���ҷ���һƿʢ����ɫ��Һ���Լ�ƿ�����ǩ���𣬴Ӳ��ಿ��ֻ�ܿ���������������Ϊ10%��������ʲô���������ϣ���ʦ����������ƿ��Һ�������������ơ��Ȼ��ơ��������ƻ���̼�����е�һ�֣�