��Ŀ����
���ý�屨����ը���е��۵�ʳƷ������ж��ı�ϩ������ ��ѧʽΪC3H5NO ����ʳ�ú��к��������Լ��� (Ҫ��д��������̣�д�ڵ�6ҳ�ϣ�ע������ʽ��) ��
��1����ϩ��������Է��������Ƕ��٣�
��2����ϩ�����и�Ԫ�ص�������m��C�� ��m��H�� ��m��N�� ��m��O���Ƕ��٣�
��3����ϩ������̼Ԫ�ص����������Ƕ��٣���������ȷ��0.1%����
��4��71g��ϩ��������ٿ� CO2������̼Ԫ�ص�������ȣ�
��1����ϩ��������Է��������Ƕ��٣�
��2����ϩ�����и�Ԫ�ص�������m��C�� ��m��H�� ��m��N�� ��m��O���Ƕ��٣�
��3����ϩ������̼Ԫ�ص����������Ƕ��٣���������ȷ��0.1%����
��4��71g��ϩ��������ٿ� CO2������̼Ԫ�ص�������ȣ�
��1��71 ��2��36��5��14��16 ��3��50.7% ��4��132g
����������⣺(1) C3H5NO����Է�������Ϊ12��3+1��5+14��1+16��1=71
(2) C3H5NO��m��C�� ��m��H�� ��m��N�� ��m��O��=��12��3������1��5������14��1������16��1��
=36��5��14��16
(3) C3H5NO��̼Ԫ�ص���������Ϊ
 ��100%
��100%=
 ��100%=50.7%
��100%=50.7%�𣺱�ϩ������̼Ԫ�ص�����������50.7%��
(4)��CO2������Ϊx
C3H5NO - 3C - 3CO2
71 132
71g x
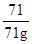 =
=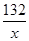
x=132g
��71g��ϩ������132�� CO2������̼Ԫ�ص��������
���������⿼��ѧ�����û�ѧʽ�ļ��㣬ѧ��Ӧץס��Ϣ�����ʵĻ�ѧʽ������ԭ�Ӹ��������ԭ��������Ԫ���غ�������㡣

��ϰ��ϵ�д�
�����Ŀ

 Na2CO3+H2O
Na2CO3+H2O
 2CaSO4 ��ijú�������������� 560 t���������Ͽ����ն���������ٶ֣�
2CaSO4 ��ijú�������������� 560 t���������Ͽ����ն���������ٶ֣�