��Ŀ����
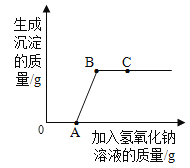
����Ŀ���ҹ���ѧ�Һ�°����˺����Ƽ��Ϊ�����Ƽҵ������ͻ��������ʵ�������IJ�Ʒ̼�����л�����������Ȼ��ƣ����и���Ʒ�����20.4g���������ྻ���ձ��У���������ε���7.3%��ϡ���ᣬ�ų�����������μ�ϡ���������Ĺ�ϵ��ͼ��ʾ����ش��������⣺

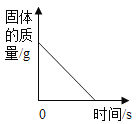
��1�����μ�ϡ������ͼ��B��ʱ���ձ�����Һ��pH_____7���<����>����=����
��2������Ʒ�������̼���Ƶ�����_____g��
��3�����μ�ϡ������ͼ��A��ʱ���ձ���������ҺΪ��������Һ��������ʱ��Һ�е�����������������д��������̣�������������0.1%��
���𰸡���1��<����2��10.6����3��18.5%
��������
��1����ͼ��֪��̼������ϡ���ᷴӦ�����Ȼ��ơ�������̼��ˮ�����μ�ϡ������ͼ��B��ʱ��ϡ�����������ʱ��Һ�е�pH��7��
��2���⣺�����Ʒ�������̼���Ƶ�����Ϊx����Ӧ�����Ȼ��Ƶ�����Ϊy����Ӧ���ɶ�����̼������Ϊz

![]() x=10.6g
x=10.6g
![]() y=11.7g
y=11.7g
![]() z=4.4g
z=4.4g
�𣺸���Ʒ�������̼���Ƶ�����Ϊ10.6g��
��3������������m��NaCl��=20.4g-10.6g=9.8g
��ʱ��Һ�е�������������Ϊ
![]()
��������������Ϊ18.5%��

 ��ѧ�̸̳����¿α�ϵ�д�
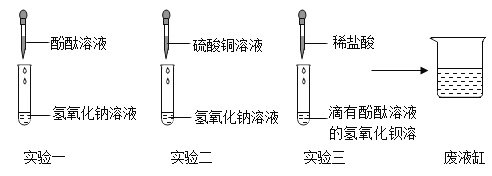
��ѧ�̸̳����¿α�ϵ�д�����Ŀ������4��ͼ���У�����ȷ��ӳ��Ӧ�仯��ϵ����
|
|
|
|
A������һ�������ĸ�����ع��� | B��ij�¶��£����������Һ�м�������ع��� | C����һ��������ϡ�����м������� | D����һ�������Ķ��������м������������Һ |
A.AB.BC.CD.D
����Ŀ��NaCl����Ҫ�Ĺ�ҵԭ�ϣ���ҵ�Ͽ��õ�ⱥ��ʳ��ˮ�ķ�����ȡ�ռ���������������ʡ�ʵ��С���ͬѧ������һ�ݾ��õĹ�ҵ�ռ�ijɷֽ���̽����
���������ϣ���ⱥ��ʳ��ˮ�Ļ�ѧ����ʽΪ��2NaCl+2H2O![]() 2NaOH+Cl2��+H2����
2NaOH+Cl2��+H2����
��������⣩С��ͬѧ���������Ϣ���Թ�ҵ�ռ�ijɷ��������²��룺
����һ��NaOH��NaCl
�������Na2CO3��NaCl
��������_________________��
��ʵ��̽����
Ϊ����֤���룬С��ͬѧ������ʵ��̽��������ȡ��ҵ�ռ���Ʒ�����������Һ�����á�
��1��ȡ��Ʒ��Һ�������μ�����ϡ���ᣬ�����ݲ�����˵����Ʒ�к���________���ѧʽ����д���������ݵĻ�ѧ��Ӧ�ķ���ʽ_________________����ҵ�ռ��к��и����ʵ�ԭ����________���û�ѧ����ʽ��ʾ����
��2��Ϊ��֤����Ʒ�к���NaCl��С��ͬѧ���ʵ�鷽�����£���ȡ��Ʒ��Һ�����������еμ�������ϡ���������ٲ�������ʱ��������Һ�еμ�AgNO3��Һ�����ְ�ɫ����������ϡ����������ܽ⣬����Ʒ��һ�������Ȼ��ơ���ʦָ��������������������˵������_____��
��3��С��ͬѧΪ��һ��ȷ����Ʒ�Ƿ������Ȼ��ƣ����������ʵ�鷽�������㽫�䲹��������
ʵ��������� | ʵ������ | ʵ����� |
��ȡ��Ʒ��Һ���������������Ȼ�����Һ,��ַ�Ӧ���� ��ȡ��Ӧ�����Һ����,�μ���ɫ��̪��Һ | ��___________ ��____________ | ����Ʒ��һ������NaOH |
����˼�����ۣ�
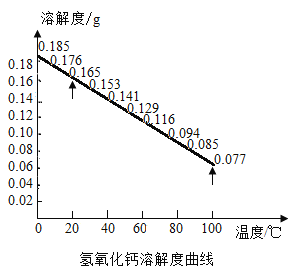
�����о���С��ͬѧ��Ϊ�����������У�����ʹ���Ȼ������⣬������������Ҳ�ɵõ�ͬ����Ч��������Ϊ������Щ�Լ��ǿ��е�_________������ţ���
A Ca��OH��2 B Ca��NO3��2 C Ba��NO3��2 D Ba��OH��2
�����ݴ�����
����ʵ��֤ʵ���þ��ù�ҵ�ռ���Ʒ�к����������ơ��Ȼ��ƺ�̼���ơ�Ϊ�˽�һ��ȷ���û������Na2CO3����������,ʵ��С���ͬѧȡ��10�˸ù�ҵ�ռ���Ʒ������������������������Ϊ7.3%ϡ���ᣬʵ��������CO2�������������������������ϵ��ͼ��ʾ�����ͼ���㣺
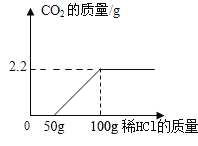
��1������Ʒ��ϡ������ȫ��Ӧʱ����CO2��������____g��
��2������Ʒ��Na2CO3����������____��